Basal late sodium current is a significant contributor to the duration of action potential of guinea pig ventricular myocytes
- PMID: 28554967
- PMCID: PMC5449569
- DOI: 10.14814/phy2.13295
Basal late sodium current is a significant contributor to the duration of action potential of guinea pig ventricular myocytes
Abstract
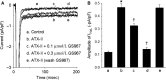
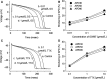
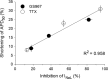
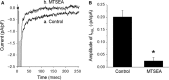
In cardiac myocytes, an enhancement of late sodium current (INaL) under pathological conditions is known to cause prolongation of action potential duration (APD). This study investigated the contribution of INaL under basal, physiological conditions to the APD Whole-cell INaL and the APD of ventricular myocytes isolated from healthy adult guinea pigs were measured at 36°C. The INaL inhibitor GS967 or TTX was applied to block INaL The amplitude of basal INaL and the APD at 50% repolarization in myocytes stimulated at a frequency of 0.17 Hz were -0.24 ± 0.02 pA/pF and 229 ± 6 msec, respectively. GS967 (0.01-1 μmol/L) concentration dependently reduced the basal INaL by 18 ± 3-82 ± 4%. At the same concentrations, GS967 shortened the APD by 9 ± 2 to 25 ± 1%. Similarly, TTX at 0.1-10 μmol/L decreased the basal INaL by 13 ± 1-94 ± 1% and APD by 8 ± 1-31 ± 2%. There was a close correlation (R2 = 0.958) between the percentage inhibition of INaL and the percentage shortening of APD caused by either GS967 or TTX MTSEA (methanethiosulfonate ethylammonium, 2 mmol/L), a NaV1.5 channel blocker, reduced the INaL by 90 ± 5%, suggesting that the NaV1.5 channel isoform is the major contributor to the basal INaL KN-93 (10 μmol/L) and AIP (2 μmol/L), blockers of CaMKII, moderately reduced the basal INaL Thus, this study provides strong evidence that basal endogenous INaL is a significant contributor to the APD of cardiac myocytes. In addition, the basal INaL of guinea pig ventricular myocytes is mainly generated from NaV1.5 channel isoform and is regulated by CaMKII.
Keywords: Action potential duration; Ca2+/calmodulin‐dependent protein kinase II; NaV1.5 channel; late sodium current; ventricular myocytes.
© 2017 The Authors. Physiological Reports published by Wiley Periodicals, Inc. on behalf of The Physiological Society and the American Physiological Society.
Conflict of interest statement
Y. Song received a research grant from Gilead Sciences; L. Belardinelli was an employee of Gilead Sciences at the time of this study.
Figures


Similar articles
-
Enhanced basal late sodium current appears to underlie the age-related prolongation of action potential duration in guinea pig ventricular myocytes.J Appl Physiol (1985). 2018 Oct 1;125(4):1329-1338. doi: 10.1152/japplphysiol.00916.2017. Epub 2017 Dec 14. J Appl Physiol (1985). 2018. PMID: 29357519
-
A computational modelling approach combined with cellular electrophysiology data provides insights into the therapeutic benefit of targeting the late Na+ current.J Physiol. 2015 Mar 15;593(6):1429-42. doi: 10.1113/jphysiol.2014.279554. J Physiol. 2015. PMID: 25545172 Free PMC article.
-
Contribution of sodium channel neuronal isoform Nav1.1 to late sodium current in ventricular myocytes from failing hearts.J Physiol. 2015 Mar 15;593(6):1409-27. doi: 10.1113/jphysiol.2014.278259. Epub 2014 Nov 17. J Physiol. 2015. PMID: 25772296 Free PMC article.
-
Late sodium current associated cardiac electrophysiological and mechanical dysfunction.Pflugers Arch. 2018 Mar;470(3):461-469. doi: 10.1007/s00424-017-2079-7. Epub 2017 Nov 10. Pflugers Arch. 2018. PMID: 29127493 Review.
-
Late sodium current in failing heart: friend or foe?Prog Biophys Mol Biol. 2008 Jan-Apr;96(1-3):421-51. doi: 10.1016/j.pbiomolbio.2007.07.010. Epub 2007 Aug 10. Prog Biophys Mol Biol. 2008. PMID: 17854868 Free PMC article. Review.
Cited by
-
Curcumin, a Multi-Ion Channel Blocker That Preferentially Blocks Late Na+ Current and Prevents I/R-Induced Arrhythmias.Front Physiol. 2020 Aug 21;11:978. doi: 10.3389/fphys.2020.00978. eCollection 2020. Front Physiol. 2020. PMID: 32973546 Free PMC article.
-
Effects of Electrical Remodeling on Atrial Fibrillation in Diabetes Mellitus.Rev Cardiovasc Med. 2023 Jan 3;24(1):3. doi: 10.31083/j.rcm2401003. eCollection 2023 Jan. Rev Cardiovasc Med. 2023. PMID: 39076858 Free PMC article. Review.
-
Late Sodium Current of the Heart: Where Do We Stand and Where Are We Going?Pharmaceuticals (Basel). 2022 Feb 15;15(2):231. doi: 10.3390/ph15020231. Pharmaceuticals (Basel). 2022. PMID: 35215342 Free PMC article. Review.
-
Inhibition of the INa/K and the activation of peak INa contribute to the arrhythmogenic effects of aconitine and mesaconitine in guinea pigs.Acta Pharmacol Sin. 2021 Feb;42(2):218-229. doi: 10.1038/s41401-020-0467-6. Epub 2020 Aug 3. Acta Pharmacol Sin. 2021. PMID: 32747718 Free PMC article.
-
Atrial Fibrillation in Diabetes: Pathogenesis and Targeted Rhythm Control Strategies.Curr Issues Mol Biol. 2025 Jul 17;47(7):559. doi: 10.3390/cimb47070559. Curr Issues Mol Biol. 2025. PMID: 40729028 Free PMC article. Review.
References
-
- Belardinelli, L. , Liu G., Smith‐Maxwell C., Wang W.‐Q., El‐Bizri N., Hirakawa R., et al. 2013. A novel, potent, and selective inhibitor of cardiac late sodium current suppresses experimental arrhythmias. J. Pharmacol. Exp. Ther. 344:23–32. - PubMed
-
- Belardinelli, L. , Giles W. R., Rajamani S., Karagueuzian H. S., and Shryock J. C.. 2015. Cardiac late Na+ current: proarrhythmic effects, roles in long QT syndromes, and pathological relationship to CaMKII and oxidative stress. Heart Rhythm 12:440–448. - PubMed
-
- Coraboeuf, E. , Deroubaix E., and Coulombe A.. 1979. Effect of tetrodotoxin on action potentials of the conducting system in the dog heart. Am. J. Physiol. 236:H561–H567. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

