Efficacy and Safety of a Chewable Methylphenidate Extended-Release Tablet in Children with Attention-Deficit/Hyperactivity Disorder
- PMID: 28557548
- PMCID: PMC5651935
- DOI: 10.1089/cap.2016.0177
Efficacy and Safety of a Chewable Methylphenidate Extended-Release Tablet in Children with Attention-Deficit/Hyperactivity Disorder
Abstract
Objective: This phase 3, laboratory classroom study assessed the efficacy and safety of methylphenidate hydrochloride extended-release chewable tablets (MPH ERCT) compared with placebo in children with attention-deficit/hyperactivity disorder (ADHD).
Methods: Following a 6-week, open-label, dose-optimization period, children 6-12 years of age (n = 90) with ADHD were randomly assigned to double-blind MPH ERCT at the final optimized dose (20-60 mg/day) or placebo. After 1 week of double-blind treatment, efficacy was assessed predose and 0.75, 2, 4, 8, 10, 12, and 13 hours postdose in a laboratory classroom setting. The primary efficacy measure was the average of postdose Swanson, Kotkin, Agler, M-Flynn, and Pelham (SKAMP) Rating Scale-Combined scores, analyzed using a mixed-model, repeated-measures analysis. Secondary efficacy measures included Permanent Product Measure of Performance (PERMP) total number of problems attempted and total number of problems correct. Safety assessments included adverse event (AE) monitoring and the Columbia-Suicide Severity Rating Scale (C-SSRS).
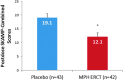
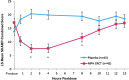
Results: MPH ERCT treatment statistically significantly reduced the average of all postdose SKAMP-Combined scores versus placebo (least-squares mean difference [95% confidence interval], -7.0 [-10.9, -3.1]; p < 0.001). Statistically significant treatment differences in SKAMP-Combined scores were observed at 2 hours postdose through 8 hours postdose (p-values <0.001). Statistically significant differences between MPH ERCT and placebo in PERMP total number of problems attempted and total number of problems correct were observed at 0.75 hours postdose through 8 hours postdose (p-values ≤0.049). Common AEs in the open-label period (≥5%) were decreased appetite, upper abdominal pain, mood swings, irritability, insomnia, upper respiratory tract infection (URTI), dysgeusia, and headache; URTI was the only AE reported by >1 subject receiving MPH ERCT in the double-blind period (placebo: URTI, contusion, wound, and initial insomnia). No suicidal ideation or behavior was reported on the C-SSRS at baseline or at any postbaseline assessment.
Conclusions: MPH ERCT 20-60 mg significantly improved ADHD symptoms compared with placebo at 2 hours postdose through at least 8 hours postdose. MPH ERCT was generally safe and well tolerated, with a safety profile consistent with other MPH ER formulations. ClinicalTrials.gov Identifier: NCT01654250. www.clinicaltrials.gov/ct2/show/NCT01654250 .
Keywords: attention-deficit/hyperactivity disorder; children; drug formulation; methylphenidate; safety; treatment efficacy.
Conflict of interest statement
Sharon Wigal receives or has received research support, acted as a consultant, been an advisory board member, and/or served on a speakers bureau for Addrenex Pharmaceuticals, Eli Lilly and Company, Forest Pharmaceuticals, McNeil Consumer & Specialty Pharmaceuticals, NextWave Pharmaceuticals, NIMH, Noven, NuTec Pharma, Otsuka, Pfizer, Purdue, Quintiles, Rho, Rhodes Pharmaceuticals, Shionogi, Shire US, Sunovion, Taisho Pharmaceutical, and Tris Pharma. Ann Childress receives or has received research support, acted as a consultant, been an advisory board member, and/or served as a speaker for Shire Pharmaceuticals, Pearson, Pfizer, Noven, NextWave Pharmaceuticals, Lilly USA, Forest Research Institute, Otsuka, Sunovion, Ironshore, Rhodes, Theravance, Neurovance, Neos, Arbor, Tris Pharma, Purdue, Lundbeck, and Alcobra. Sally A. Berry is an employee of Tris Pharma and has served as a paid consultant for Pfizer in connection with the development of the manuscript. Heidi Belden was a paid consultant for Pfizer in connection with the development of this manuscript. Faith Walters, Phillip Chappell, Nancy A. Sherman, John Orazem, and Donna Palumbo are employees of Pfizer.
Figures
References
-
- Abbas R, Palumbo D, Walters F, Belden H, Berry SA: Single-dose pharmacokinetics and relative bioavailability of a novel methylphenidate extended-release chewable tablet compared with immediate-release methylphenidate chewable tablet. Clin Ther 38:1151–1157, 2016 - PubMed
-
- Adjei A, Teuscher NS, Kupper RJ, Chang WW, Greenhill L, Newcorn JH, Connor DF, Wigal S: Single-dose pharmacokinetics of methylphenidate extended-release multiple layer beads administered as intact capsule or sprinkles versus methylphenidate immediate-release tablets (Ritalin((R))) in healthy adult volunteers. J Child Adolesc Psychopharmacol 24:570–578, 2014 - PMC - PubMed
-
- American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013
-
- Aptensio XR. [package insert]: Coventry (Rhode Island), Rhodes Pharmaceuticals, 2017
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical