Axonal Conduction Delays, Brain State, and Corticogeniculate Communication
- PMID: 28559382
- PMCID: PMC5490068
- DOI: 10.1523/JNEUROSCI.0444-17.2017
Axonal Conduction Delays, Brain State, and Corticogeniculate Communication
Abstract
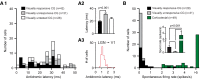
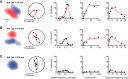
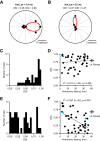
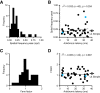
Thalamocortical conduction times are short, but layer 6 corticothalamic axons display an enormous range of conduction times, some exceeding 40-50 ms. Here, we investigate (1) how axonal conduction times of corticogeniculate (CG) neurons are related to the visual information conveyed to the thalamus, and (2) how alert versus nonalert awake brain states affect visual processing across the spectrum of CG conduction times. In awake female Dutch-Belted rabbits, we found 58% of CG neurons to be visually responsive, and 42% to be unresponsive. All responsive CG neurons had simple, orientation-selective receptive fields, and generated sustained responses to stationary stimuli. CG axonal conduction times were strongly related to modulated firing rates (F1 values) generated by drifting grating stimuli, and their associated interspike interval distributions, suggesting a continuum of visual responsiveness spanning the spectrum of axonal conduction times. CG conduction times were also significantly related to visual response latency, contrast sensitivity (C-50 values), directional selectivity, and optimal stimulus velocity. Increasing alertness did not cause visually unresponsive CG neurons to become responsive and did not change the response linearity (F1/F0 ratios) of visually responsive CG neurons. However, for visually responsive CG neurons, increased alertness nearly doubled the modulated response amplitude to optimal visual stimulation (F1 values), significantly shortened response latency, and dramatically increased response reliability. These effects of alertness were uniform across the broad spectrum of CG axonal conduction times.SIGNIFICANCE STATEMENT Corticothalamic neurons of layer 6 send a dense feedback projection to thalamic nuclei that provide input to sensory neocortex. While sensory information reaches the cortex after brief thalamocortical axonal delays, corticothalamic axons can exhibit conduction delays of <2 ms to 40-50 ms. Here, in the corticogeniculate visual system of awake rabbits, we investigate the functional significance of this axonal diversity, and the effects of shifting alert/nonalert brain states on corticogeniculate processing. We show that axonal conduction times are strongly related to multiple visual response properties, suggesting a continuum of visual responsiveness spanning the spectrum of corticogeniculate axonal conduction times. We also show that transitions between awake brain states powerfully affect corticogeniculate processing, in some ways more strongly than in layer 4.
Keywords: alert brain state; corticothalamic; lateral geniculate nucleus; layer 6; neocortical axons; visual cortex.
Copyright © 2017 the authors 0270-6474/17/376342-17$15.00/0.
Conflict of interest statement
The authors declare no competing financial interests.
Figures









Similar articles
-
Corticogeniculate neurons, corticotectal neurons, and suspected interneurons in visual cortex of awake rabbits: receptive-field properties, axonal properties, and effects of EEG arousal.J Neurophysiol. 1987 Apr;57(4):977-1001. doi: 10.1152/jn.1987.57.4.977. J Neurophysiol. 1987. PMID: 3585466
-
Efferent neurons and suspected interneurons in binocular visual cortex of the awake rabbit: receptive fields and binocular properties.J Neurophysiol. 1988 Apr;59(4):1162-87. doi: 10.1152/jn.1988.59.4.1162. J Neurophysiol. 1988. PMID: 3373273
-
Getting drowsy? Alert/nonalert transitions and visual thalamocortical network dynamics.J Neurosci. 2011 Nov 30;31(48):17480-7. doi: 10.1523/JNEUROSCI.2262-11.2011. J Neurosci. 2011. PMID: 22131409 Free PMC article.
-
How much feedback from visual cortex to lateral geniculate nucleus in cat: a perspective.Vis Neurosci. 2004 Jul-Aug;21(4):487-500. doi: 10.1017/S0952523804214018. Vis Neurosci. 2004. PMID: 15579216 Review.
-
Corticothalamic interactions in the transfer of visual information.Philos Trans R Soc Lond B Biol Sci. 2002 Dec 29;357(1428):1739-52. doi: 10.1098/rstb.2002.1170. Philos Trans R Soc Lond B Biol Sci. 2002. PMID: 12626008 Free PMC article. Review.
Cited by
-
The synaptic inputs and thalamic projections of two classes of layer 6 corticothalamic neurons in primary somatosensory cortex of the mouse.J Comp Neurol. 2021 Dec;529(17):3751-3771. doi: 10.1002/cne.25163. Epub 2021 May 6. J Comp Neurol. 2021. PMID: 33908623 Free PMC article.
-
Role of Feedback Connections in Central Visual Processing.Annu Rev Vis Sci. 2020 Sep 15;6:313-334. doi: 10.1146/annurev-vision-121219-081716. Epub 2020 Jun 17. Annu Rev Vis Sci. 2020. PMID: 32552571 Free PMC article. Review.
-
Imaging high-frequency voltage dynamics in multiple neuron classes of behaving mammals.bioRxiv [Preprint]. 2024 Aug 16:2024.08.15.607428. doi: 10.1101/2024.08.15.607428. bioRxiv. 2024. Update in: Cell. 2025 Aug 7;188(16):4401-4423.e31. doi: 10.1016/j.cell.2025.06.028. PMID: 39185175 Free PMC article. Updated. Preprint.
-
Dendritic and Axonal Propagation Delays May Shape Neuronal Networks With Plastic Synapses.Front Physiol. 2018 Dec 20;9:1849. doi: 10.3389/fphys.2018.01849. eCollection 2018. Front Physiol. 2018. PMID: 30618847 Free PMC article. Review.
-
A Closer Look at Corticothalamic "Loops".Front Neural Circuits. 2021 Feb 2;15:632668. doi: 10.3389/fncir.2021.632668. eCollection 2021. Front Neural Circuits. 2021. PMID: 33603649 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
