Achiote (Bixa orellana L.): a natural source of pigment and vitamin E
- PMID: 28559632
- PMCID: PMC5430180
- DOI: 10.1007/s13197-017-2579-7
Achiote (Bixa orellana L.): a natural source of pigment and vitamin E
Abstract
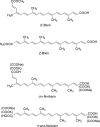
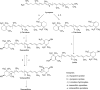
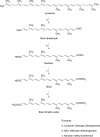
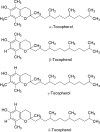
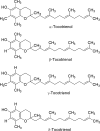
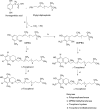
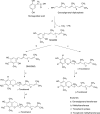
Commercialization of agricultural products, including seeds and its derived products, represents an important economic source for developing countries. Natural colorants obtained from the seeds of achiote plant (annatto) have been used since pre-Hispanic times. Also, production of this crop has been important for Mayan cuisine. Annual world production of achiote seeds is approximately 14,500 tons (dry weight). Two thirds of the production is commercialized as dried seeds and the rest as colorant. Latin America produces 60% of the total world production, followed by Africa (27%) and Asia (12%). The main producers in Latin America are Peru, Brazil and Mexico. The purpose of the present paper is to review the most recent literature on Bixa orellana L. focusing on bixin, norbixin, tocotrienols and tocopherols biosynthesis, use and industrial applications of annatto extracts, as well as its nutraceutical potential and its benefits for human health.
Keywords: Annatto extract; Bixin; Norbixin; Tocopherols; Tocotrienols.
Figures
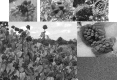
References
-
- Cardarelli CR, Benassi MT, Mercadante AZ. Characterization of different annatto extracts based on antioxidant and colour properties. LWT-Food Sci Technol. 2008;41:1689–1693. doi: 10.1016/j.lwt.2007.10.013. - DOI
-
- Castro CV, Murcia SCL. Licopeno y Salud Humana. Rev Reciteia. 2013;12:42–79.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
