Synthesis and Preliminary Studies of a Novel Negative Allosteric Modulator, 7-((2,5-Dioxopyrrolidin-1-yl)methyl)-4-(2-fluoro-4-[11C]methoxyphenyl) quinoline-2-carboxamide, for Imaging of Metabotropic Glutamate Receptor 2
- PMID: 28565908
- PMCID: PMC5607115
- DOI: 10.1021/acschemneuro.7b00098
Synthesis and Preliminary Studies of a Novel Negative Allosteric Modulator, 7-((2,5-Dioxopyrrolidin-1-yl)methyl)-4-(2-fluoro-4-[11C]methoxyphenyl) quinoline-2-carboxamide, for Imaging of Metabotropic Glutamate Receptor 2
Abstract
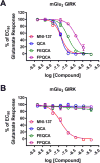
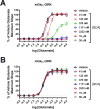
Metabotropic glutamate 2 receptors (mGlu2) are involved in the pathogenesis of several CNS disorders and neurodegenerative diseases. Pharmacological modulation of this target represents a potential disease-modifying approach for the treatment of substance abuse, depression, schizophrenia, and dementias. While quantification of mGlu2 receptors in the living brain by positron emission tomography (PET) would help us better understand signaling pathways relevant to these conditions, few successful examples have been demonstrated to image mGlu2 in vivo, and a suitable PET tracer is yet to be identified. Herein we report the design and synthesis of a radiolabeled negative allosteric modulator (NAM) for mGlu2 PET tracer development based on a quinoline 2-carboxamide scaffold. The most promising candidate, 7-((2,5-dioxopyrrolidin-1-yl)methyl)-4-(2-fluoro-4-[11C]methoxyphenyl) quinoline-2-carboxamide ([11C]QCA) was prepared in 13% radiochemical yield (non-decay-corrected at the end of synthesis) with >99% radiochemical purity and >74 GBq/μmol (2 Ci/μmol) specific activity. While the tracer showed limited brain uptake (0.3 SUV), probably attributable to effects on PgP/Bcrp efflux pump, in vitro autoradiography studies demonstrated heterogeneous brain distribution and specific binding. Thus, [11C]QCA is a chemical probe that provides the basis for the development of a new generation mGlu2 PET tracers.
Keywords: 11C; Positron emission tomography; mGlu2; metabotropic glutamate receptor 2; negative allosteric modulator.
Figures

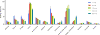
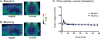

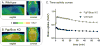
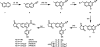
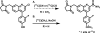
Similar articles
-
Identification and Development of a New Positron Emission Tomography Ligand 4-(2-Fluoro-4-[11C]methoxyphenyl)-5-((1-methyl-1H-pyrazol-3-yl)methoxy)picolinamide for Imaging Metabotropic Glutamate Receptor Subtype 2 (mGlu2).J Med Chem. 2020 Oct 22;63(20):11469-11483. doi: 10.1021/acs.jmedchem.9b01991. Epub 2020 Sep 22. J Med Chem. 2020. PMID: 32960052 Free PMC article.
-
Synthesis and preliminary studies of 11C-labeled tetrahydro-1,7-naphthyridine-2-carboxamides for PET imaging of metabotropic glutamate receptor 2.Theranostics. 2020 Sep 14;10(24):11178-11196. doi: 10.7150/thno.42587. eCollection 2020. Theranostics. 2020. PMID: 33042277 Free PMC article.
-
Synthesis and evaluation of 4-(2-fluoro-4-[11C]methoxyphenyl)-5-((2-methylpyridin-4-yl)methoxy)picolinamide for PET imaging of the metabotropic glutamate receptor 2 in the rat brain.Bioorg Med Chem. 2019 Feb 1;27(3):483-491. doi: 10.1016/j.bmc.2018.12.025. Epub 2018 Dec 17. Bioorg Med Chem. 2019. PMID: 30611634
-
Development of allosteric modulators of GPCRs for treatment of CNS disorders.Neurobiol Dis. 2014 Jan;61:55-71. doi: 10.1016/j.nbd.2013.09.013. Epub 2013 Sep 27. Neurobiol Dis. 2014. PMID: 24076101 Free PMC article. Review.
-
Pharmacological and molecular characterization of the positive allosteric modulators of metabotropic glutamate receptor 2.Neuropharmacology. 2016 Dec;111:253-265. doi: 10.1016/j.neuropharm.2016.08.032. Epub 2016 Aug 30. Neuropharmacology. 2016. PMID: 27590915 Review.
Cited by
-
Synthesis and Characterization of 5-(2-Fluoro-4-[11C]methoxyphenyl)-2,2-dimethyl-3,4-dihydro-2H-pyrano[2,3-b]pyridine-7-carboxamide as a PET Imaging Ligand for Metabotropic Glutamate Receptor 2.J Med Chem. 2022 Feb 10;65(3):2593-2609. doi: 10.1021/acs.jmedchem.1c02004. Epub 2022 Jan 28. J Med Chem. 2022. PMID: 35089713 Free PMC article.
-
Design, Synthesis, and Characterization of Benzimidazole Derivatives as Positron Emission Tomography Imaging Ligands for Metabotropic Glutamate Receptor 2.J Med Chem. 2020 Oct 22;63(20):12060-12072. doi: 10.1021/acs.jmedchem.0c01394. Epub 2020 Oct 7. J Med Chem. 2020. PMID: 32981322 Free PMC article.
-
Design and Synthesis of N-Aryl Phenoxyethoxy Pyridinones as Highly Selective and CNS Penetrant mGlu3 NAMs.ACS Med Chem Lett. 2017 Aug 15;8(9):925-930. doi: 10.1021/acsmedchemlett.7b00249. eCollection 2017 Sep 14. ACS Med Chem Lett. 2017. PMID: 28947938 Free PMC article.
-
A Review of Molecular Imaging of Glutamate Receptors.Molecules. 2020 Oct 16;25(20):4749. doi: 10.3390/molecules25204749. Molecules. 2020. PMID: 33081223 Free PMC article. Review.
-
Radiosynthesis and Evaluation of 11C-Labeled Isoindolone-Based Positive Allosteric Modulators for Positron Emission Tomography Imaging of Metabotropic Glutamate Receptor 2.ACS Pharmacol Transl Sci. 2024 Jul 10;7(8):2414-2423. doi: 10.1021/acsptsci.4c00261. eCollection 2024 Aug 9. ACS Pharmacol Transl Sci. 2024. PMID: 39144551 Free PMC article.
References
-
- Nakanishi S. Molecular diversity of glutamate receptors and implications for brain function. Science. 1992;258:597–603. - PubMed
-
- Kew JN, Kemp JA. Ionotropic and metabotropic glutamate receptor structure and pharmacology. Psychopharmacology. 2005;179:4–29. - PubMed
-
- Lau A, Tymianski M. Glutamate receptors, neurotoxicity and neurodegeneration. Pflugers Arch. 2010;460:525–542. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

