Click-chemistry approaches to π-conjugated polymers for organic electronics applications
- PMID: 28567241
- PMCID: PMC5450439
- DOI: 10.1039/c6sc01832g
Click-chemistry approaches to π-conjugated polymers for organic electronics applications
Abstract
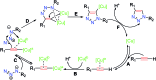
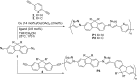
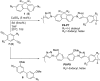
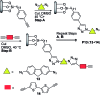
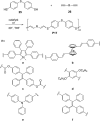
Given the wide utility of click-chemistry reactions for the preparation of simple moieties within large architecturally complex materials, this minireview article aims at surveying papers exploring their scope in the area of π-conjugated polymers for application in organic electronics to enable advanced functional properties.
Figures












Similar articles
-
Conjugated Polymers in Bioelectronics.Acc Chem Res. 2018 Jun 19;51(6):1368-1376. doi: 10.1021/acs.accounts.7b00624. Epub 2018 Jun 6. Acc Chem Res. 2018. PMID: 29874033
-
Molecular Approach to Conjugated Polymers with Biomimetic Properties.Acc Chem Res. 2018 Jul 17;51(7):1581-1589. doi: 10.1021/acs.accounts.7b00596. Epub 2018 Jun 13. Acc Chem Res. 2018. PMID: 29897228
-
Novel Conjugated Polymers Prepared by Direct (Hetero) arylation: An Eco-Friendly Tool for Organic Electronics.Molecules. 2018 Feb 13;23(2):408. doi: 10.3390/molecules23020408. Molecules. 2018. PMID: 29438329 Free PMC article.
-
Naphthobischalcogenadiazole Conjugated Polymers: Emerging Materials for Organic Electronics.Adv Mater. 2017 Jul;29(25). doi: 10.1002/adma.201605218. Epub 2017 Feb 27. Adv Mater. 2017. PMID: 28240796 Review.
-
Doping the Backbone of π-Conjugated Polymers with Tricoordinate Boron: Synthetic Strategies and Emerging Applications.Chem Asian J. 2019 Apr 1;14(7):919-935. doi: 10.1002/asia.201900016. Epub 2019 Feb 27. Chem Asian J. 2019. PMID: 30731024 Review.
Cited by
-
Effect of curvature and placement of donor and acceptor units in cycloparaphenylenes: a computational study.Chem Sci. 2020 Sep 25;11(44):12029-12035. doi: 10.1039/d0sc03923c. Chem Sci. 2020. PMID: 34094422 Free PMC article.
-
Expedient Azide-Alkyne Huisgen Cycloaddition Catalyzed by a Combination of VOSO4 with Cu(0) in Aqueous Media.ACS Org Inorg Au. 2023 Dec 19;4(2):235-240. doi: 10.1021/acsorginorgau.3c00059. eCollection 2024 Apr 3. ACS Org Inorg Au. 2023. PMID: 38585512 Free PMC article.
-
"Clickable" Organic Electrochemical Transistors.JACS Au. 2022 Nov 23;2(12):2778-2790. doi: 10.1021/jacsau.2c00515. eCollection 2022 Dec 26. JACS Au. 2022. PMID: 36590273 Free PMC article.
-
Recent Development on Narrow Bandgap Conjugated Polymers for Polymer Solar Cells.Polymers (Basel). 2017 Jan 28;9(2):39. doi: 10.3390/polym9020039. Polymers (Basel). 2017. PMID: 30970721 Free PMC article. Review.
-
Synthesis of Metalorganic Copolymers Containing Various Contorted Units and Iron(II) Clathrochelates with Lateral Butyl Chains: Conspicuous Adsorbents of Lithium Ions and Methylene Blue.Polymers (Basel). 2022 Aug 19;14(16):3394. doi: 10.3390/polym14163394. Polymers (Basel). 2022. PMID: 36015650 Free PMC article.
References
-
-
For recent representative reviews see:
- Yao Y., Dong H., Hu W. Adv. Mater. 2016;28:4513–4523. - PubMed
- Nielsen C. B., Holliday S., Chen H.-Y., Cryer S. J., McCulloch I. Acc. Chem. Res. 2015;48:2803–2812. - PMC - PubMed
- Dou L., Liu Y., Hong Z., Li G., Yang Y. Chem. Rev. 2015;115:12633–12665. - PubMed
- Muellen K., Pisula W. J. Am. Chem. Soc. 2015;137:9503–9505. - PubMed
- Yi Z., Wang S., Liu Y. Adv. Mater. 2015;27:3589–3606. - PubMed
- Savagatrup S., Printz A. D., O'Connor T. F., Zaretski A. V., Lipomi D. J. Chem. Mater. 2014;26:3028–3041.
- Benight S. J., Wang C., Tok J. B. H., Bao Z. Prog. Polym. Sci. 2013;38:1961–1977.
- Marrocchi A., Lanari D., Facchetti A., Vaccaro L. Energy Environ. Sci. 2012;5:8457–8474.
-
-
- Khikhlovskyi V., Van Bremen A. J. J., Janssen R. A. J., Gelink G. H., Kemerink M. Org. Electron. 2016;31:56–62.
- Nam S., Seo J., Kim H., Kim Y. Appl. Phys. Lett. 2015;107:153302.
- Meena J. S., Sze S. M., Chand U., Tseng T.-Y. Nanoscale Res. Lett. 2014;9:526–559. - PMC - PubMed
- Han S.-T., Zhou Y., Roy V. A. L. Adv. Mater. 2013;25:5425–5449. - PubMed
-
- Metal catalyzed cross-coupling reactions and more, ed. A. de Meijere, S. Braese and M. Oestreich, Wiley-VCH, Weinheim, Germany, 2014.
-
-
For representative examples see:
- Strappaveccia G., Ismalaj E., Petrucci C., Lanari D., Marrocchi A., Drees M., Facchetti A., Vaccaro L. Green Chem. 2015;17:365–372.
- Osedach T. P., Andrew T. L., Bulović V. Energy Environ. Sci. 2013;6:711–718.
- Leong W. L., Welch G. C., Kaake L. G., Takacs C. J., Sun Y., Bazan G. C., Heeger A. J. Chem. Sci. 2012;3:2103–2109.
-
-
-
For representative examples see:
- Morin P.-O., Bura T., Leclerc M. Mater. Horiz. 2016;3:11–20.
- Suraru S.-L., Lee J. A., Luscombe C. ACS Macro Lett. 2016;5:724–729. - PubMed
- Rudenko A. E., Thompson B. C. J. Polym. Sci., Part A: Polym. Chem. 2015;53:135–147.
- Mercier L. G., Leclerc M. Acc. Chem. Res. 2013;46:1597–1605. - PubMed
- Facchetti A., Marrocchi A., Vaccaro L. Angew. Chem., Int. Ed. 2012;51:3520–3523. - PubMed
-
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

