Coupling of excitation to Ca2+ release is modulated by dysferlin
- PMID: 28568606
- PMCID: PMC5538227
- DOI: 10.1113/JP274515
Coupling of excitation to Ca2+ release is modulated by dysferlin
Abstract
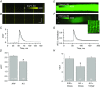
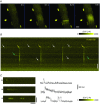
Key points: Dysferlin, the protein missing in limb girdle muscular dystrophy 2B and Miyoshi myopathy, concentrates in transverse tubules of skeletal muscle, where it stabilizes voltage-induced Ca2+ transients against loss after osmotic shock injury (OSI). Local expression of dysferlin in dysferlin-null myofibres increases transient amplitude to control levels and protects them from loss after OSI. Inhibitors of ryanodine receptors (RyR1) and L-type Ca2+ channels protect voltage-induced Ca2+ transients from loss; thus both proteins play a role in injury in dysferlin's absence. Effects of Ca2+ -free medium and S107, which inhibits SR Ca2+ leak, suggest the SR as the primary source of Ca2+ responsible for the loss of the Ca2+ transient upon injury. Ca2+ waves were induced by OSI and suppressed by exogenous dysferlin. We conclude that dysferlin prevents injury-induced SR Ca2+ leak.
Abstract: Dysferlin concentrates in the transverse tubules of skeletal muscle and stabilizes Ca2+ transients when muscle fibres are subjected to osmotic shock injury (OSI). We show here that voltage-induced Ca2+ transients elicited in dysferlin-null A/J myofibres were smaller than control A/WySnJ fibres. Regional expression of Venus-dysferlin chimeras in A/J fibres restored the full amplitude of the Ca2+ transients and protected against OSI. We also show that drugs that target ryanodine receptors (RyR1: dantrolene, tetracaine, S107) and L-type Ca2+ channels (LTCCs: nifedipine, verapamil, diltiazem) prevented the decrease in Ca2+ transients in A/J fibres following OSI. Diltiazem specifically increased transients by ∼20% in uninjured A/J fibres, restoring them to control values. The fact that both RyR1s and LTCCs were involved in OSI-induced damage suggests that damage is mediated by increased Ca2+ leak from the sarcoplasmic reticulum (SR) through the RyR1. Congruent with this, injured A/J fibres produced Ca2+ sparks and Ca2+ waves. S107 (a stabilizer of RyR1-FK506 binding protein coupling that reduces Ca2+ leak) or local expression of Venus-dysferlin prevented OSI-induced Ca2+ waves. Our data suggest that dysferlin modulates SR Ca2+ release in skeletal muscle, and that in its absence OSI causes increased RyR1-mediated Ca2+ leak from the SR into the cytoplasm.
Keywords: dysferlin; excitation-contraction coupling; skeletal muscle.
© 2017 The Authors. The Journal of Physiology © 2017 The Physiological Society.
Figures







References
-
- Aksentsev SL, Rakovich AA, Okoon IM, Konev SV, Orlov SN & Kravtsov GM (1983). Effect of tetracaine on veratrine‐mediated influx of sodium into rat brain synaptosomes. Pflugers Arch 397, 135–140. - PubMed
-
- Bannister RA (2016). Bridging the myoplasmic gap II: more recent advances in skeletal muscle excitation‐contraction coupling. J Exp Biol 219, 175–182. - PubMed
-
- Bansal D & Campbell KP (2004). Dysferlin and the plasma membrane repair in muscular dystrophy. Trends Cell Biol 14, 206–213. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

