Fibroblast-like synoviocyte metabolism in the pathogenesis of rheumatoid arthritis
- PMID: 28569176
- PMCID: PMC5452638
- DOI: 10.1186/s13075-017-1303-3
Fibroblast-like synoviocyte metabolism in the pathogenesis of rheumatoid arthritis
Abstract
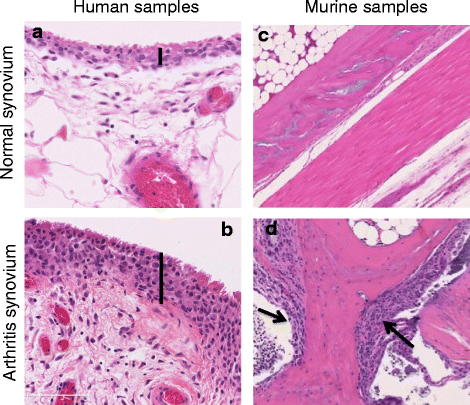
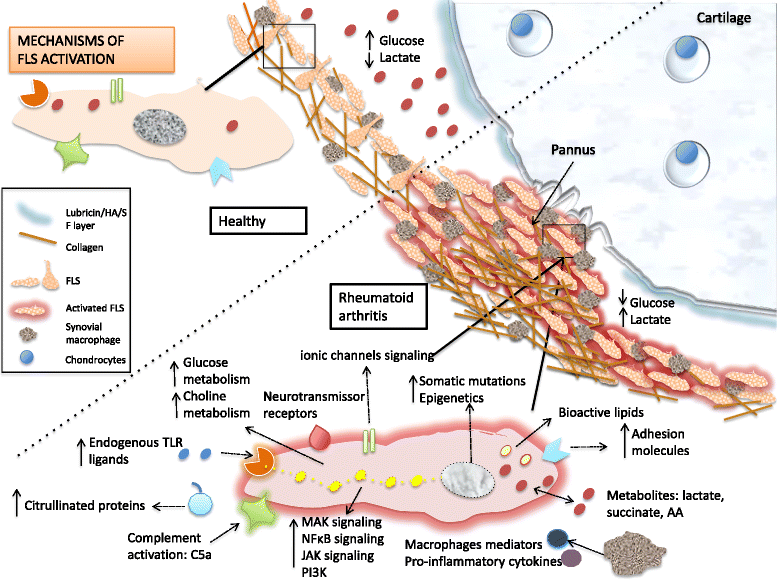
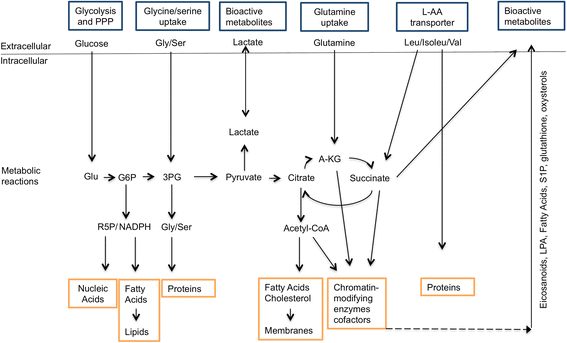
An increasing number of studies show how changes in intracellular metabolic pathways alter tumor and immune cell function. However, little information about metabolic changes in other cell types, including synovial fibroblasts, is available. In rheumatoid arthritis (RA), fibroblast-like synoviocytes (FLS) are the most common cell type at the pannus-cartilage junction and contribute to joint destruction through their production of cytokines, chemokines, and matrix-degrading molecules and by migrating and invading joint cartilage. In this review, we show that these cells differ from healthy synovial fibroblasts, not only in their marker expression, proto-oncogene expression, or their epigenetic changes, but also in their intracellular metabolism. These metabolic changes must occur due to the stressful microenvironment of inflamed tissues, where concentrations of crucial nutrients such as glucose, glutamine, and oxygen are spatially and temporally heterogeneous. In addition, these metabolic changes will increase metabolite exchange between fibroblast and other synovial cells, which can potentially be activated. Glucose and phospholipid metabolism as well as bioactive lipids, including sphingosine-1-phosphate and lysophosphatidic acid, among others, are involved in FLS activation. These metabolic changes likely contribute to FLS involvement in aspects of immune response initiation or abnormal immune responses and strongly contribute to joint destruction.
Keywords: Bioactive lipids; FLS; Fibroblast-like synovicyte; Glycolysis; Metabolism.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

