Tumour-associated macrophages secrete pleiotrophin to promote PTPRZ1 signalling in glioblastoma stem cells for tumour growth
- PMID: 28569747
- PMCID: PMC5461490
- DOI: 10.1038/ncomms15080
Tumour-associated macrophages secrete pleiotrophin to promote PTPRZ1 signalling in glioblastoma stem cells for tumour growth
Abstract
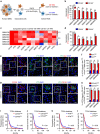
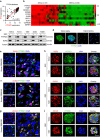
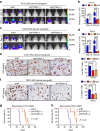
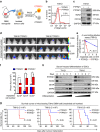
Intense infiltration of tumour-associated macrophages (TAMs) facilitates malignant growth of glioblastoma (GBM), but the underlying mechanisms remain undefined. Herein, we report that TAMs secrete abundant pleiotrophin (PTN) to stimulate glioma stem cells (GSCs) through its receptor PTPRZ1 thus promoting GBM malignant growth through PTN-PTPRZ1 paracrine signalling. PTN expression correlates with infiltration of CD11b+/CD163+ TAMs and poor prognosis of GBM patients. Co-implantation of M2-like macrophages (MLCs) promoted GSC-driven tumour growth, but silencing PTN expression in MLCs mitigated their pro-tumorigenic activity. The PTN receptor PTPRZ1 is preferentially expressed in GSCs and also predicts GBM poor prognosis. Disrupting PTPRZ1 abrogated GSC maintenance and tumorigenic potential. Moreover, blocking the PTN-PTPRZ1 signalling by shRNA or anti-PTPRZ1 antibody potently suppressed GBM tumour growth and prolonged animal survival. Our study uncovered a critical molecular crosstalk between TAMs and GSCs through the PTN-PTPRZ1 paracrine signalling to support GBM malignant growth, indicating that targeting this signalling axis may have therapeutic potential.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



References
-
- Stupp R. et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 10, 459–466 (2009). - PubMed
-
- Garris C. & Pittet M. J. Therapeutically reeducating macrophages to treat GBM. Nat. Med. 19, 1207–1208 (2013). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

