Bi-specific molecule against EGFR and death receptors simultaneously targets proliferation and death pathways in tumors
- PMID: 28572590
- PMCID: PMC5454031
- DOI: 10.1038/s41598-017-02483-9
Bi-specific molecule against EGFR and death receptors simultaneously targets proliferation and death pathways in tumors
Abstract
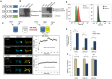
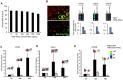
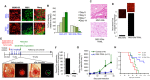
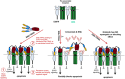
Developing therapeutics that target multiple receptor signaling pathways in tumors is critical as therapies targeting single specific biomarker/pathway have shown limited efficacy in patients with cancer. In this study, we extensively characterized a bi-functional molecule comprising of epidermal growth factor receptor (EGFR) targeted nanobody (ENb) and death receptor (DR) targeted ligand TRAIL (ENb-TRAIL). We show that ENb-TRAIL has therapeutic efficacy in tumor cells from different cancer types which do not respond to either EGFR antagonist or DR agonist monotherapies. Utilizing pharmacological inhibition, genetic loss of function and FRET studies, we show that ENb-TRAIL blocks EGFR signalling via the binding of ENb to EGFR which in turn induces DR5 clustering at the plasma membrane and thereby primes tumor cells to caspase-mediated apoptosis. In vivo, using a clinically relevant orthotopic resection model of primary glioblastoma and engineered stem cells (SC) expressing ENb-TRAIL, we show that the treatment with synthetic extracellular matrix (sECM) encapsulated SC-ENb-TRAIL alleviates tumor burden and significantly increases survival. This study is the first to report novel mechanistic insights into simultaneous targeting of receptor-mediated proliferation and cell death signaling pathways in different tumor types and presents a promising approach for translation into the clinical setting.
Conflict of interest statement
“Khalid Shah (K.S.) owns equity in and is a member of the Board of Directors of, AMASA Technologies, a company developing stem cell based therapies for cancer. KS’s interests were reviewed and are managed by Brigham and Women’s Hospital and Partners HealthCare in accordance with their conflict of interest policies”. The other authors declare that there is no conflict of interest.
Figures


Similar articles
-
Therapeutic stem cells expressing variants of EGFR-specific nanobodies have antitumor effects.Proc Natl Acad Sci U S A. 2012 Oct 9;109(41):16642-7. doi: 10.1073/pnas.1202832109. Epub 2012 Sep 25. Proc Natl Acad Sci U S A. 2012. PMID: 23012408 Free PMC article.
-
A bispecific fusion protein and a bifunctional enediyne-energized fusion protein consisting of TRAIL, EGFR peptide ligand, and apoprotein of lidamycin against EGFR and DR4/5 show potent antitumor activity.Anticancer Drugs. 2015 Jan;26(1):64-73. doi: 10.1097/CAD.0000000000000160. Anticancer Drugs. 2015. PMID: 25122444
-
Bortezomib primes glioblastoma, including glioblastoma stem cells, for TRAIL by increasing tBid stability and mitochondrial apoptosis.Clin Cancer Res. 2011 Jun 15;17(12):4019-30. doi: 10.1158/1078-0432.CCR-11-0075. Epub 2011 Apr 27. Clin Cancer Res. 2011. PMID: 21525171
-
TRAIL receptor signalling and modulation: Are we on the right TRAIL?Cancer Treat Rev. 2009 May;35(3):280-8. doi: 10.1016/j.ctrv.2008.11.006. Epub 2008 Dec 30. Cancer Treat Rev. 2009. PMID: 19117685 Review.
-
TRAIL receptor signaling and therapeutics.Expert Opin Ther Targets. 2010 Oct;14(10):1091-108. doi: 10.1517/14728222.2010.519701. Expert Opin Ther Targets. 2010. PMID: 20819019 Review.
Cited by
-
Single-Domain Antibodies and the Promise of Modular Targeting in Cancer Imaging and Treatment.Front Immunol. 2018 Feb 19;9:273. doi: 10.3389/fimmu.2018.00273. eCollection 2018. Front Immunol. 2018. PMID: 29520274 Free PMC article. Review.
-
Glioblastoma cells vampirize WNT from neurons and trigger a JNK/MMP signaling loop that enhances glioblastoma progression and neurodegeneration.PLoS Biol. 2019 Dec 17;17(12):e3000545. doi: 10.1371/journal.pbio.3000545. eCollection 2019 Dec. PLoS Biol. 2019. PMID: 31846454 Free PMC article.
-
Molecular Mode of Action of TRAIL Receptor Agonists-Common Principles and Their Translational Exploitation.Cancers (Basel). 2019 Jul 7;11(7):954. doi: 10.3390/cancers11070954. Cancers (Basel). 2019. PMID: 31284696 Free PMC article. Review.
-
Developing and characterizing a two-layered safety switch for cell therapies.Cancer Biol Ther. 2023 Dec 31;24(1):2232146. doi: 10.1080/15384047.2023.2232146. Cancer Biol Ther. 2023. PMID: 37439774 Free PMC article.
-
Axl and EGFR Dual-Specific Binding Affibody for Targeted Therapy in Nasopharyngeal Carcinoma.Cells. 2024 Nov 5;13(22):1823. doi: 10.3390/cells13221823. Cells. 2024. PMID: 39594573 Free PMC article.
References
-
- Shinojima N, et al. Prognostic value of epidermal growth factor receptor in patients with glioblastoma multiforme. Cancer research. 2003;63:6962–6970. - PubMed
-
- Thatcher N, et al. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: results from a randomised, placebo-controlled, multicentre study (Iressa Survival Evaluation in Lung Cancer) Lancet. 2005;366:1527–1537. doi: 10.1016/S0140-6736(05)67625-8. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

