A New Adjuvant MTOM Mediates Mycobacterium tuberculosis Subunit Vaccine to Enhance Th1-Type T Cell Immune Responses and IL-2+ T Cells
- PMID: 28572807
- PMCID: PMC5436497
- DOI: 10.3389/fimmu.2017.00585
A New Adjuvant MTOM Mediates Mycobacterium tuberculosis Subunit Vaccine to Enhance Th1-Type T Cell Immune Responses and IL-2+ T Cells
Abstract
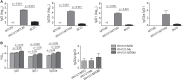
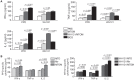
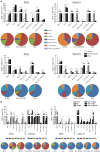
The only licensed vaccine Mycobacterium bovis Bacillus Calmette-Guérin (BCG) cannot prevent the prevalence of tuberculosis (TB), which remains a major public health problem worldwide. A more effective TB vaccine than BCG is urgently needed. Subunit vaccine is a promising strategy, and suitable adjuvants will benefit the development of effective TB subunit vaccines. MTO, consisting of monophosphoryl lipid A (MPLA), trehalose-6,6'-dibehenate (TDB), and MF59, was developed as an adjuvant of TB vaccine because of its ability to evoke the Th1-type T cell responses, while it is insufficient to induce single and multifunctional IL-2+ T cells and has a limited ability to confer protection against Mycobacterium tuberculosis infection. Heat-killed Mycobacterium vaccae (Mv), which can evoke cytotoxic CD8+ and CD4+ T cell responses and has adjuvanticity, was, in this study, combined with MTO to produce a new adjuvant, called MTOM. The TB fusion protein Rv3407-PhoY2-Ag85A-Rv2626c-RpfB (WH121) was mixed with MTO, Mv, and MTOM to produce three subunit vaccines, and the protective efficacy and immune responses were compared in C57BL/6 mice. WH121/MTOM provided better protection against TB than the other two vaccines, matching the performance of BCG vaccine. MTOM showed stronger ability to increase single and multifunctional IL-2+ T cells and induce Th1-type responses than MTO or Mv. Therefore, MTOM might be a promising adjuvant that could contribute to the development of TB subunit vaccines.
Keywords: IL-2+ T cells; MTOM; Mycobacterium tuberculosis; Th1-type response; adjuvant.
Figures

References
-
- Global Tuberculosis Report 2016. Geneva, Switzerland: United Nations, World Health Organization (WHO) (2016). Available from: http://www.who.int/tb/publications/global_report/en/
-
- Yao S, Huang D, Chen CY, Halliday L, Wang RC, Chen ZW. CD4+ T cells contain early extrapulmonary tuberculosis (TB) dissemination and rapid TB progression and sustain multieffector functions of CD8+ T and CD3- lymphocytes: mechanisms of CD4+ T cell immunity. J Immunol (2014) 192(5):2120–32. 10.4049/jimmunol.1301373 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

