Efficacy of digital pupillometry for diagnosis of Horner syndrome
- PMID: 28575101
- PMCID: PMC5456040
- DOI: 10.1371/journal.pone.0178361
Efficacy of digital pupillometry for diagnosis of Horner syndrome
Abstract
Objectives: To evaluate the efficacy of digital pupillometry in the diagnosis of anisocoria related to Horner syndrome in adult patients.
Design: Retrospective, observational, case control study.
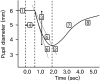
Methods: Nineteen patients with unilateral Horner syndrome (Horner group) and age-matched controls of 30 healthy individuals with normal vision and neither optic nerve dysfunction nor pupillary abnormalities were included. Pupillary light reflex (PLR) of the Horner group and controls were measured by a dynamic pupillometer (PLR-200; NeurOptics Inc., Irvine, USA). Minimal and maximal (min/max) pupil diameters, latency, constriction ratio, constriction velocity, dilation velocity, and total time taken by the pupil to recover 75% of maximal pupil diameter (T75) were noted. PLR were measured at baseline in both groups and at 30-45 minutes later after 0.5% apraclonidine (Iopidine®; Alcon Laboratories, Fort Worth, TX, USA) instillation in the Horner group.
Main outcome measures: The PLR parameters in the affected eye and inter-eye difference before and after 0.5% apraclonidine instillation.
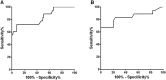
Results: In the Horner group, pupil diameters and T75 showed significant difference between the affected eye and unaffected contralateral eye at baseline (all P<0.00625). Compared to controls, inter-eye difference values of pupil diameters and T75 were significantly larger in the Horner group (all P<0.001). After 0.5% apraclonidine instillation, changes in pupil diameter and constriction ratio were significantly larger in the affected eye compared to the unaffected contralateral eye (all P<0.00625). The area under the receiver operating characteristic curves for diagnosing Horner syndrome were largest for baseline inter-eye difference in min/max pupil sizes (AUC = 0.975, 0.994), T75 (AUC = 0.838), and change in min/max pupil sizes after apraclonidine instillation (AUC = 0.923, 0.929, respectively). The diagnostic criteria for Horner syndrome relying on baseline pupillary measurements was defined as one of the two major findings; 1) smaller maximal pupil diameter in the affected eye with an inter-eye difference of > 0.5 mm, or 2) T75 > 2.61 seconds in the affected eye, which showed a sensitivity of 94.7% and specificity of 93.3%. The diagnostic accuracy of apraclonidine testing showed a sensitivity of 84.6% and specificity of 92.3%.
Conclusions: Digital pupillometry is an objective method for quantifying PLR. Baseline inter-eye difference in maximal pupil sizes and dilation lag measured by T75 was equally effective in the diagnosis of Horner syndrome compared to the reversal of anisocoria after apraclonidine instillation.
Conflict of interest statement
Figures

References
-
- Langham ME, Weinstein GW. Horner's syndrome. Ocular supersensitivity to adrenergic amines. Arch Ophthalmol. 1967;78(4):462–469. . - PubMed
-
- Walton KA, Buono LM. Horner syndrome. Curr Opin Ophthalmol. 2003;14(6):357–363. . - PubMed
-
- Kardon RH, Denison CE, Brown CK, Thompson HS. Critical evaluation of the cocaine test in the diagnosis of Horner's syndrome. Arch Ophthalmol. 1990;108(3):384–387. . - PubMed
-
- Mughal M, Longmuir R. Current pharmacologic testing for Horner syndrome. Curr Neurol Neurosci Rep. 2009;9(5):384–389. . - PubMed
-
- Abrams DA, Robin AL, Pollack IP, deFaller JM, DeSantis L. The safety and efficacy of topical 1% ALO 2145 (p-aminoclonidine hydrochloride) in normal volunteers. Arch Ophthalmol. 1987;105(9):1205–1207. . - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

