Alginate microbeads are coagulation compatible, while alginate microcapsules activate coagulation secondary to complement or directly through FXII
- PMID: 28576714
- PMCID: PMC5607061
- DOI: 10.1016/j.actbio.2017.05.052
Alginate microbeads are coagulation compatible, while alginate microcapsules activate coagulation secondary to complement or directly through FXII
Abstract
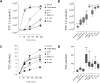
Alginate microspheres are presently under evaluation for future cell-based therapy. Their ability to induce harmful host reactions needs to be identified for developing the most suitable devices and efficient prevention strategies. We used a lepirudin based human whole blood model to investigate the coagulation potentials of alginate-based microspheres: alginate microbeads (Ca/Ba Beads), alginate poly-l-lysine microcapsules (APA and AP microcapsules) and sodium alginate-sodium cellulose sulfate-poly(methylene-co-cyanoguanidine) microcapsules (PMCG microcapsules). Coagulation activation measured by prothrombin fragments 1+2 (PTF1.2) was rapidly and markedly induced by the PMCG microcapsules, delayed and lower induced by the APA and AP microcapsules, and not induced by the Ca/Ba Beads. Monocytes tissue factor (TF) expression was similarly activated by the microcapsules, whereas not by the Ca/Ba Beads. PMCG microcapsules-induced PTF1.2 was abolished by FXII inhibition (corn trypsin inhibitor), thus pointing to activation through the contact pathway. PTF1.2 induced by the AP and APA microcapsules was inhibited by anti-TF antibody, pointing to a TF driven coagulation. The TF induced coagulation was inhibited by the complement inhibitors compstatin (C3 inhibition) and eculizumab (C5 inhibition), revealing a complement-coagulation cross-talk. This is the first study on the coagulation potentials of alginate microspheres, and identifies differences in activation potential, pathways and possible intervention points.
Statement of significance: Alginate microcapsules are prospective candidate materials for cell encapsulation therapy. The material surface must be free of host cell adhesion to ensure free diffusion of nutrition and oxygen to the encapsulated cells. Coagulation activation is one gateway to cellular overgrowth through deposition of fibrin. Herein we used a physiologically relevant whole blood model to investigate the coagulation potential of alginate microcapsules and microbeads. The coagulation potentials and the pathways of activation were depending on the surface properties of the materials. Activation of the complement system could also be involved, thus emphasizing a complement-coagulation cross-talk. Our findings points to complement and coagulation inhibition as intervention point for preventing host reactions, and enhance functional cell-encapsulation devices.
Keywords: Alginate microcapsules; Coagulation; Complement; Cross-talk; Factor XII; Tissue factor.
Copyright © 2017 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.
Conflict of interest statement
The authors declare no conflicts of interests. J.D.L. is the inventor of patents and/or patent applications that describe the use of complement inhibitors for therapeutic purposes and the founder of Amyndas Pharmaceuticals, which is developing complement inhibitors for clinical applications. We confirm that all authors have approved the final article.
Figures
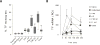





Similar articles
-
Alginate microsphere compositions dictate different mechanisms of complement activation with consequences for cytokine release and leukocyte activation.J Control Release. 2016 May 10;229:58-69. doi: 10.1016/j.jconrel.2016.03.021. Epub 2016 Mar 16. J Control Release. 2016. PMID: 26993426
-
Sulfated alginate microspheres associate with factor H and dampen the inflammatory cytokine response.Acta Biomater. 2016 Sep 15;42:180-188. doi: 10.1016/j.actbio.2016.06.015. Epub 2016 Jun 11. Acta Biomater. 2016. PMID: 27296843
-
Alginate microbeads are complement compatible, in contrast to polycation containing microcapsules, as revealed in a human whole blood model.Acta Biomater. 2011 Jun;7(6):2566-78. doi: 10.1016/j.actbio.2011.03.011. Epub 2011 Mar 12. Acta Biomater. 2011. PMID: 21402181
-
Alginate-based microcapsules generated with the coaxial electrospray method for clinical application.J Biomater Sci Polym Ed. 2017 Sep;28(13):1245-1255. doi: 10.1080/09205063.2017.1318030. Epub 2017 Apr 20. J Biomater Sci Polym Ed. 2017. PMID: 28391767 Review.
-
Sulfated Alginates as Heparin Analogues: A Review of Chemical and Functional Properties.Molecules. 2017 May 11;22(5):778. doi: 10.3390/molecules22050778. Molecules. 2017. PMID: 28492485 Free PMC article. Review.
Cited by
-
MS-proteomics provides insight into the host responses towards alginate microspheres.Mater Today Bio. 2022 Nov 11;17:100490. doi: 10.1016/j.mtbio.2022.100490. eCollection 2022 Dec 15. Mater Today Bio. 2022. PMID: 36420052 Free PMC article.
-
Cholesterol Crystals Induce Coagulation Activation through Complement-Dependent Expression of Monocytic Tissue Factor.J Immunol. 2019 Aug 15;203(4):853-863. doi: 10.4049/jimmunol.1900503. Epub 2019 Jul 3. J Immunol. 2019. PMID: 31270150 Free PMC article.
-
Low-Dose Aspirin Treatment Attenuates Male Rat Salt-Sensitive Hypertension via Platelet Cyclooxygenase 1 and Complement Cascade Pathway.J Am Heart Assoc. 2020 Jan 7;9(1):e013470. doi: 10.1161/JAHA.119.013470. Epub 2019 Dec 19. J Am Heart Assoc. 2020. PMID: 31852420 Free PMC article.
-
Fucoidan alginate and sulfated alginate microbeads induce distinct coagulation, inflammatory and fibrotic responses.Mater Today Bio. 2025 Jan 10;31:101474. doi: 10.1016/j.mtbio.2025.101474. eCollection 2025 Apr. Mater Today Bio. 2025. PMID: 39896282 Free PMC article.
-
Type 1 diabetes mellitus (T1DM) does not affect whole blood responses to alginate-based microspheres despite plasma lipid and glucose differences.Mater Today Bio. 2025 Jul 18;34:102113. doi: 10.1016/j.mtbio.2025.102113. eCollection 2025 Oct. Mater Today Bio. 2025. PMID: 40755902 Free PMC article.
References
-
- Rokstad AM, Lacik I, de Vos P, Strand BL. Advances in biocompatibility and physicochemical characterization of microspheres for cell encapsulation. Advanced drug delivery reviews. 2014;67–68:111–30. - PubMed
-
- Rokstad AM, Brekke OL, Steinkjer B, Ryan L, Kollarikova G, Strand BL, Skjak-Braek G, Lacik I, Espevik T, Mollnes TE. Alginate microbeads are complement compatible, in contrast to polycation containing microcapsules, as revealed in a human whole blood model. Acta Biomater. 2011;7(6):2566–2578. - PubMed
-
- Orning P, Hoem KS, Coron AE, Skjak-Braek G, Mollnes TE, Brekke OL, Espevik T, Rokstad AM. Alginate microsphere compositions dictate different mechanisms of complement activation with consequences for cytokine release and leukocyte activation. J Control Release. 2016;229:58–69. - PubMed
-
- Rokstad AM, Brekke OL, Steinkjer B, Ryan L, Kollarikova G, Strand BL, Skjak-Braek G, Lambris JD, Lacik I, Mollnes TE, Espevik T. The induction of cytokines by polycation containing microspheres by a complement dependent mechanism 1. Biomaterials. 2013;34(3):621–630. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

