The type II isopentenyl Diphosphate:Dimethylallyl diphosphate isomerase (IDI-2): A model for acid/base chemistry in flavoenzyme catalysis
- PMID: 28577910
- PMCID: PMC5650523
- DOI: 10.1016/j.abb.2017.05.017
The type II isopentenyl Diphosphate:Dimethylallyl diphosphate isomerase (IDI-2): A model for acid/base chemistry in flavoenzyme catalysis
Abstract
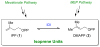
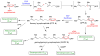
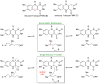
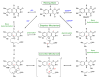
The chemical versatility of the flavin coenzyme is nearly unparalleled in enzyme catalysis. An interesting illustration of this versatility can be found in the reaction catalyzed by the type II isopentenyl diphosphate:dimethylallyl diphosphate isomerase (IDI-2) - an enzyme that interconverts the two essential isoprene units (isopentenyl pyrophosphate and dimethylallyl pyrophosphate) that are needed to initiate the biosynthesis of all isoprenoids. Over the past decade, a variety of biochemical, spectroscopic, structural and mechanistic studies of IDI-2 have provided mounting evidence that the flavin coenzyme of IDI-2 acts in a most unusual manner - as an acid/base catalyst to mediate a 1,3-proton addition/elimination reaction. While not entirely without precedent, IDI-2 is by far the most extensively studied flavoenzyme that employs flavin-mediated acid/base catalysis. Thus, IDI-2 serves as an important mechanistic model for understanding this often overlooked, but potentially widespread reactivity of flavin coenzymes. This review details the most pertinent studies that have contributed to the development of mechanistic proposals for this highly unusual flavoenzyme, and discusses future experiments that may be able to clarify remaining uncertainties in the chemical mechanism of IDI-2.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures





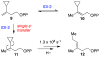



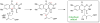

Similar articles
-
Linear free energy relationships demonstrate a catalytic role for the flavin mononucleotide coenzyme of the type II isopentenyl diphosphate:dimethylallyl diphosphate isomerase.J Am Chem Soc. 2010 Jul 28;132(29):9994-6. doi: 10.1021/ja104090m. J Am Chem Soc. 2010. PMID: 20593767 Free PMC article.
-
Kinetic and Binding Studies of Streptococcus pneumoniae Type 2 Isopentenyl Diphosphate:Dimethylallyl Diphosphate Isomerase.Biochemistry. 2016 Apr 19;55(15):2260-8. doi: 10.1021/acs.biochem.6b00087. Epub 2016 Apr 5. Biochemistry. 2016. PMID: 27003727
-
Type-2 isopentenyl diphosphate isomerase: evidence for a stepwise mechanism.J Am Chem Soc. 2011 Nov 30;133(47):19017-9. doi: 10.1021/ja208331q. Epub 2011 Nov 9. J Am Chem Soc. 2011. PMID: 22047048 Free PMC article.
-
Isopentenyl diphosphate isomerase: A checkpoint to isoprenoid biosynthesis.Biochimie. 2012 Aug;94(8):1621-34. doi: 10.1016/j.biochi.2012.03.021. Epub 2012 Apr 4. Biochimie. 2012. PMID: 22503704 Review.
-
Diversity of the biosynthesis of the isoprene units.Nat Prod Rep. 2003 Apr;20(2):171-83. doi: 10.1039/b109860h. Nat Prod Rep. 2003. PMID: 12735695 Review.
Cited by
-
The subcellular localization of two isopentenyl diphosphate isomerases in rice suggests a role for the endoplasmic reticulum in isoprenoid biosynthesis.Plant Cell Rep. 2020 Jan;39(1):119-133. doi: 10.1007/s00299-019-02479-x. Epub 2019 Nov 2. Plant Cell Rep. 2020. PMID: 31679061
-
Identification and a phased pH control strategy of diosgenin bio-synthesized by an endogenous Bacillus licheniformis Syt1 derived from Dioscorea zingiberensis C. H. Wright.Appl Microbiol Biotechnol. 2021 Dec;105(24):9333-9342. doi: 10.1007/s00253-021-11679-z. Epub 2021 Nov 29. Appl Microbiol Biotechnol. 2021. PMID: 34841464
-
Engineering plant family TPS into cyanobacterial host for terpenoids production.Plant Cell Rep. 2022 Sep;41(9):1791-1803. doi: 10.1007/s00299-022-02892-9. Epub 2022 Jul 5. Plant Cell Rep. 2022. PMID: 35789422 Free PMC article. Review.
References
-
- Porter JW, Spurgeon SL. In: Biosynthesis of isoprenoid compounds. Porter JW, Spurgeon SL, editors. John Wiley and Sons; New York: 1981. pp. 1–93.
-
- Bloch K, Chaykin S, Phillips AH, de Waard A. J Biol Chem. 1959;234:2595–2604. - PubMed
-
- Lynen F, Eggerer H, Henning U, Kessel I. Angew Chem. 1958;70:738–742.
-
- Beytia ED, Porter JW. Annu Rev Biochem. 1976;45:113–142. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

