Paradoxical roles of dual oxidases in cancer biology
- PMID: 28578013
- PMCID: PMC5535817
- DOI: 10.1016/j.freeradbiomed.2017.05.024
Paradoxical roles of dual oxidases in cancer biology
Abstract
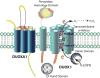
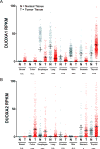
Dysregulated oxidative metabolism is a well-recognized aspect of cancer biology, and many therapeutic strategies are based on targeting cancers by altering cellular redox pathways. The NADPH oxidases (NOXes) present an important enzymatic source of biological oxidants, and the expression and activation of several NOX isoforms are frequently dysregulated in many cancers. Cell-based studies have demonstrated a role for several NOX isozymes in controlling cell proliferation and/or cell migration, further supporting a potential contributing role for NOX in promoting cancer. While various NOX isoforms are often upregulated in cancers, paradoxical recent findings indicate that dual oxidases (DUOXes), normally prominently expressed in epithelial lineages, are frequently suppressed in epithelial-derived cancers by epigenetic mechanisms, although the functional relevance of such DUOX silencing has remained unclear. This review will briefly summarize our current understanding regarding the importance of reactive oxygen species (ROS) and NOXes in cancer biology, and focus on recent observations indicating the unique and seemingly opposing roles of DUOX enzymes in cancer biology. We will discuss current knowledge regarding the functional properties of DUOX, and recent studies highlighting mechanistic consequences of DUOX1 loss in lung cancer, and its consequences for tumor invasiveness and current anticancer therapy. Finally, we will also discuss potentially unique roles for the DUOX maturation factors. Overall, a better understanding of mechanisms that regulate DUOX and the functional consequences of DUOX silencing in cancer may offer valuable new diagnostic insights and novel therapeutic opportunities.
Keywords: DUOX; EGFR; Epigenetics; Epithelial; NADPH oxidase; NOX; Reactive oxygen species.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
DUOX1 in mammalian disease pathophysiology.J Mol Med (Berl). 2021 Jun;99(6):743-754. doi: 10.1007/s00109-021-02058-2. Epub 2021 Mar 11. J Mol Med (Berl). 2021. PMID: 33704512 Free PMC article. Review.
-
The Dual Role of Reactive Oxygen Species-Generating Nicotinamide Adenine Dinucleotide Phosphate Oxidases in Gastrointestinal Inflammation and Therapeutic Perspectives.Antioxid Redox Signal. 2020 Aug 10;33(5):354-373. doi: 10.1089/ars.2020.8018. Epub 2020 Feb 26. Antioxid Redox Signal. 2020. PMID: 31968991
-
Antimicrobial actions of dual oxidases and lactoperoxidase.J Microbiol. 2018 Jun;56(6):373-386. doi: 10.1007/s12275-018-7545-1. Epub 2018 Jun 1. J Microbiol. 2018. PMID: 29858825 Free PMC article. Review.
-
Molecular evolution of the reactive oxygen-generating NADPH oxidase (Nox/Duox) family of enzymes.BMC Evol Biol. 2007 Jul 6;7:109. doi: 10.1186/1471-2148-7-109. BMC Evol Biol. 2007. PMID: 17612411 Free PMC article.
-
Functional Characterization of DUOX Enzymes in Reconstituted Cell Models.Methods Mol Biol. 2019;1982:173-190. doi: 10.1007/978-1-4939-9424-3_11. Methods Mol Biol. 2019. PMID: 31172473
Cited by
-
Transcriptomic analysis reveals oxidative stress-related signature and molecular subtypes in cholangio carcinoma.Mol Genet Genomics. 2024 Sep 6;299(1):86. doi: 10.1007/s00438-024-02170-1. Mol Genet Genomics. 2024. PMID: 39240371
-
Insights on Localized and Systemic Delivery of Redox-Based Therapeutics.Oxid Med Cell Longev. 2018 Feb 14;2018:2468457. doi: 10.1155/2018/2468457. eCollection 2018. Oxid Med Cell Longev. 2018. PMID: 29636836 Free PMC article. Review.
-
Peroxidasin-mediated crosslinking of collagen IV is independent of NADPH oxidases.Redox Biol. 2018 Jun;16:314-321. doi: 10.1016/j.redox.2018.03.009. Epub 2018 Mar 14. Redox Biol. 2018. PMID: 29573705 Free PMC article.
-
Exploring redox vulnerabilities in JAK2V617F-positive cellular models.Hematol Transfus Cell Ther. 2021 Oct-Dec;43(4):430-436. doi: 10.1016/j.htct.2020.08.006. Epub 2020 Sep 13. Hematol Transfus Cell Ther. 2021. PMID: 32962959 Free PMC article.
-
Molecular characterization of AwSox2 from bivalve Anodonta woodiana: Elucidating its player in the immune response.Innate Immun. 2020 Jul;26(5):381-397. doi: 10.1177/1753425919897823. Epub 2019 Dec 31. Innate Immun. 2020. PMID: 31889462 Free PMC article.
References
-
- Lambeth JD. NOX enzymes and the biology of reactive oxygen. Nature reviews. Immunology. 2004;4(3):181–9. - PubMed
-
- Lambeth JD, Neish AS. Nox enzymes and new thinking on reactive oxygen: a double-edged sword revisited. Annual review of pathology. 2014;9:119–45. - PubMed
-
- Holmstrom KM, Finkel T. Cellular mechanisms and physiological consequences of redox-dependent signalling. Nat Rev Mol Cell Biol. 2014;15(6):411–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

