Abiraterone for Prostate Cancer Not Previously Treated with Hormone Therapy
- PMID: 28578639
- PMCID: PMC5533216
- DOI: 10.1056/NEJMoa1702900
Abiraterone for Prostate Cancer Not Previously Treated with Hormone Therapy
Abstract
Background: Abiraterone acetate plus prednisolone improves survival in men with relapsed prostate cancer. We assessed the effect of this combination in men starting long-term androgen-deprivation therapy (ADT), using a multigroup, multistage trial design.
Methods: We randomly assigned patients in a 1:1 ratio to receive ADT alone or ADT plus abiraterone acetate (1000 mg daily) and prednisolone (5 mg daily) (combination therapy). Local radiotherapy was mandated for patients with node-negative, nonmetastatic disease and encouraged for those with positive nodes. For patients with nonmetastatic disease with no radiotherapy planned and for patients with metastatic disease, treatment continued until radiologic, clinical, or prostate-specific antigen (PSA) progression; otherwise, treatment was to continue for 2 years or until any type of progression, whichever came first. The primary outcome measure was overall survival. The intermediate primary outcome was failure-free survival (treatment failure was defined as radiologic, clinical, or PSA progression or death from prostate cancer).
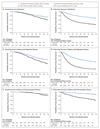
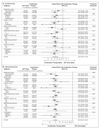
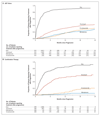
Results: A total of 1917 patients underwent randomization from November 2011 through January 2014. The median age was 67 years, and the median PSA level was 53 ng per milliliter. A total of 52% of the patients had metastatic disease, 20% had node-positive or node-indeterminate nonmetastatic disease, and 28% had node-negative, nonmetastatic disease; 95% had newly diagnosed disease. The median follow-up was 40 months. There were 184 deaths in the combination group as compared with 262 in the ADT-alone group (hazard ratio, 0.63; 95% confidence interval [CI], 0.52 to 0.76; P<0.001); the hazard ratio was 0.75 in patients with nonmetastatic disease and 0.61 in those with metastatic disease. There were 248 treatment-failure events in the combination group as compared with 535 in the ADT-alone group (hazard ratio, 0.29; 95% CI, 0.25 to 0.34; P<0.001); the hazard ratio was 0.21 in patients with nonmetastatic disease and 0.31 in those with metastatic disease. Grade 3 to 5 adverse events occurred in 47% of the patients in the combination group (with nine grade 5 events) and in 33% of the patients in the ADT-alone group (with three grade 5 events).
Conclusions: Among men with locally advanced or metastatic prostate cancer, ADT plus abiraterone and prednisolone was associated with significantly higher rates of overall and failure-free survival than ADT alone. (Funded by Cancer Research U.K. and others; STAMPEDE ClinicalTrials.gov number, NCT00268476 , and Current Controlled Trials number, ISRCTN78818544 .).
Figures
Comment in
-
Prostate cancer: STAMPEDE, LATITUDE and Fernand Labrie's legacy.Nat Rev Urol. 2017 Oct;14(10):588-590. doi: 10.1038/nrurol.2017.115. Epub 2017 Jul 18. Nat Rev Urol. 2017. PMID: 28720861 No abstract available.
-
Improved Outcomes in Men with Advanced Prostate Cancer.N Engl J Med. 2017 Jul 27;377(4):388-390. doi: 10.1056/NEJMe1704992. N Engl J Med. 2017. PMID: 28745978 No abstract available.
-
Expanding the use of abiraterone in prostate cancer: Is earlier always better?Cancer Biol Ther. 2018 Feb 1;19(2):97-100. doi: 10.1080/15384047.2017.1394555. Epub 2017 Dec 8. Cancer Biol Ther. 2018. PMID: 29219670 Free PMC article. No abstract available.
-
[Improved survival in advanced or metastatic prostate cancer with initial androgen deprivation and concomitant abiraterone].Strahlenther Onkol. 2018 Mar;194(3):266-268. doi: 10.1007/s00066-017-1252-4. Strahlenther Onkol. 2018. PMID: 29302703 German. No abstract available.
-
Re: Abiraterone for Prostate Cancer Not Previously Treated with Hormone Therapy.Eur Urol. 2018 Jun;73(6):981. doi: 10.1016/j.eururo.2018.01.023. Epub 2018 Feb 9. Eur Urol. 2018. PMID: 29433974 No abstract available.
References
-
- Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411–22. - PubMed
-
- Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–12. - PubMed
-
- Petrylak DP, Tangen CM, Hussain MHA, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004;351:1513–20. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
