Cell-Cycle Therapeutics Come of Age
- PMID: 28580868
- PMCID: PMC6075824
- DOI: 10.1200/JCO.2016.69.0032
Cell-Cycle Therapeutics Come of Age
Abstract
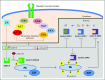
The ability to sustain unscheduled proliferation is a hallmark of cancer. The normal process of cell division occurs via the cell cycle, a series of highly regulated steps that are orchestrated at the molecular level by specific cyclins that act in association with cyclin-dependent kinases (CDKs). Cyclin D and CDK4/6 play a key role in cell-cycle progression by phosphorylating and inactivating the retinoblastoma protein, a tumor suppressor that restrains G1- to S-phase progression. The first-generation CDK inhibitors demonstrated broad activity upon several CDKs, which likely explains their considerable toxicities and limited efficacy. Palbociclib, ribociclib, and abemaciclib represent a new class of highly specific ATP-competitive CDK4/6 inhibitors that induce reversible G1-phase cell-cycle arrest in retinoblastoma-positive tumor models. Both palbociclib and ribociclib have been approved in combination with hormone-based therapy for the treatment of naïve hormone receptor-positive advanced breast cancer on the basis of an improvement in progression-free survival. In general, CDK4/6 inhibitors are cytostatic as monotherapy but demonstrate favorable tolerability, which has prompted interest in combination approaches. Combinations with phosphatidylinositol 3-kinase and mammalian target of rapamycin inhibitors in breast cancer, and inhibitors of the RAS/RAF/mitogen-activated protein kinase pathway in RAS-mutant cancers are particularly promising approaches that are currently being evaluated. Although the subject of intense preclinical study, predictive biomarkers for response and resistance to these drugs remain largely undefined. CDK4/6 inhibitors have emerged as the most promising of the cell-cycle therapeutics and intense efforts are now underway to expand the reach of this paradigm.
Figures
References
-
- Hanahan D, Weinberg RA: Hallmarks of cancer: The next generation. Cell 144:646-674, 2011 - PubMed
-
- Shapiro GI: Cyclin-dependent kinase pathways as targets for cancer treatment. J Clin Oncol 24:1770-1783, 2006 - PubMed
-
- Nurse PM: Nobel lecture. Cyclin dependent kinases and cell cycle control. Biosci Rep 22:487-499, 2002 - PubMed
-
- Weinberg RA: The Biology of Cancer. New York, NY, Garland Science, 2014
-
- Sicinski P, Donaher JL, Parker SB, et al. : Cyclin D1 provides a link between development and oncogenesis in the retina and breast. Cell 82:621-630, 1995 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

