High quality clinical grade human embryonic stem cell lines derived from fresh discarded embryos
- PMID: 28583200
- PMCID: PMC5460457
- DOI: 10.1186/s13287-017-0561-y
High quality clinical grade human embryonic stem cell lines derived from fresh discarded embryos
Abstract
Background: Human embryonic stem cells (hESCs) hold tremendous promise for cell replacement therapies for a range of degenerative diseases. In order to provide cost-effective treatments affordable by public health systems, HLA-matched allogeneic tissue banks of the highest quality clinical-grade hESCs will be required. However only a small number of existing hESC lines are suitable for clinical use; they are limited by moral and ethical concerns and none of them apply Good Manufacturing Practice (GMP) standards to the earliest and critical stages of gamete and embryo procurement. We thus aimed to derive new clinical grade hESC lines of highest quality from fresh surplus GMP grade human embryos.
Methods: A comprehensive screen was performed for suitable combinations of culture media with supporting feeder cells or feeder-free matrix, at different stages, to support expansion of the inner cell mass and to establish new hESC lines.
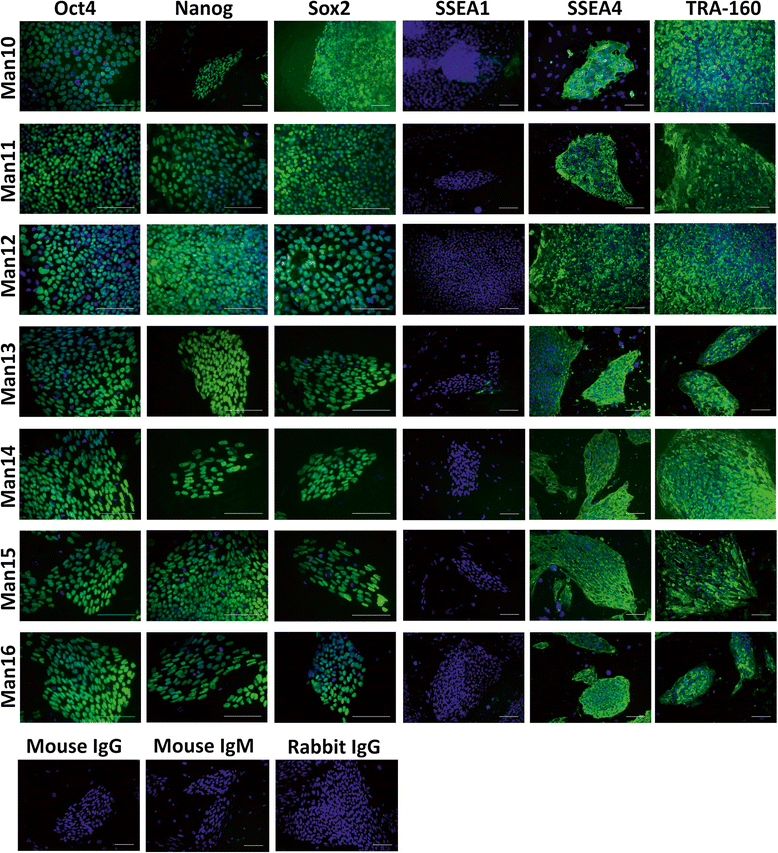
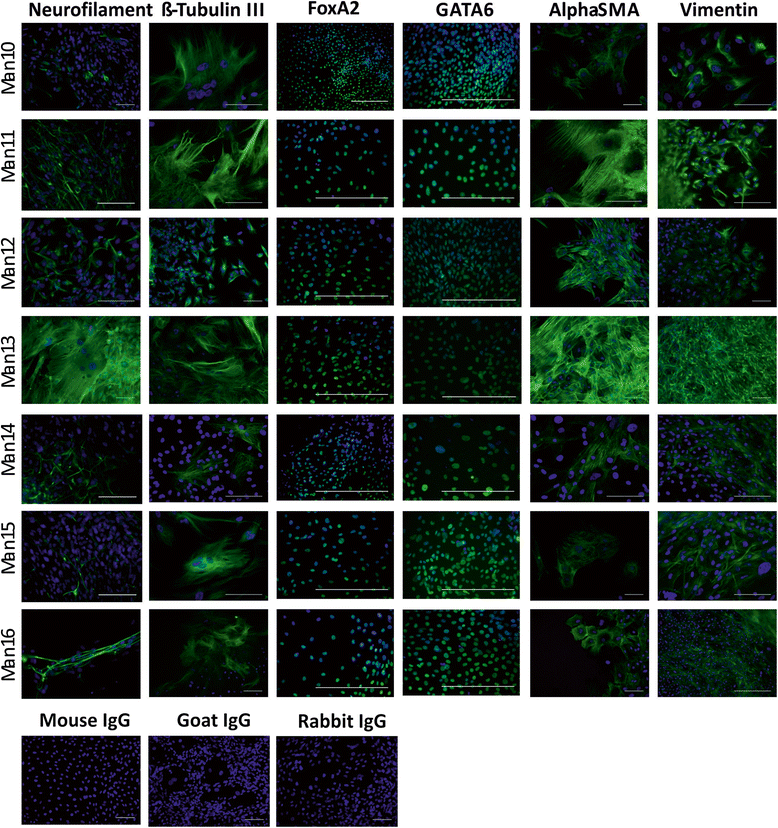
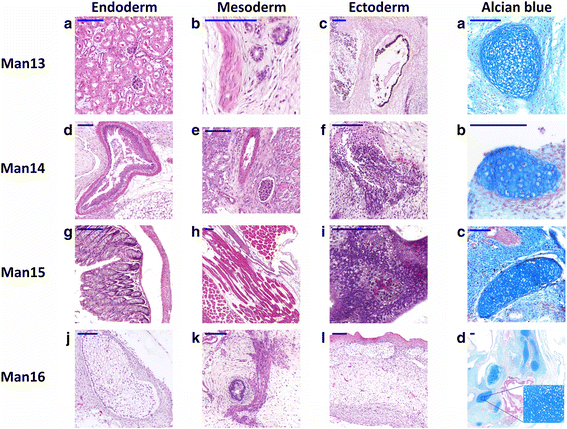
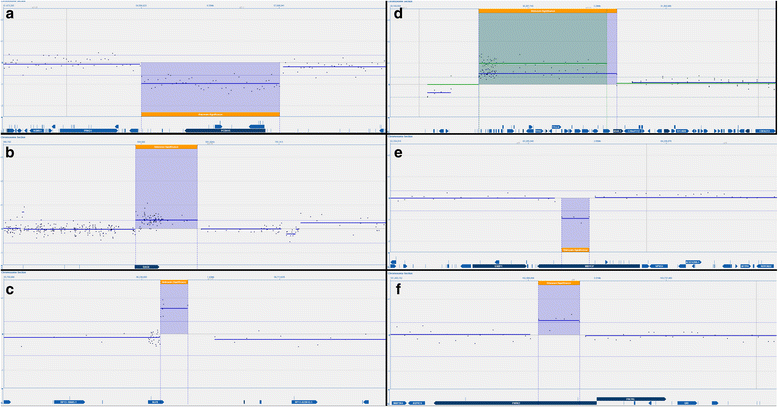
Results: We developed a novel two-step and sequential media system of clinical-grade hESC derivation and successfully generated seven new hESC lines of widely varying HLA type, carefully screened for genetic health, from human embryos donated under the highest ethical and moral standards under an integrated GMP system which extends from hESC banking all the way back to gamete and embryo procurement.
Conclusions: The present study, for the first time, reports the successful derivation of highest-quality clinical-grade hESC lines from fresh poor-quality surplus human embryos generated in a GMP-grade IVF laboratory. The availability of hESC lines of this status represents an important step towards more widespread application of regenerative medicine therapies.
Keywords: Embryo; Good Manufacturing Practice; Human embryonic stem cells; Pluripotency.
Figures

References
-
- Arabadjiev B, Petkova R, Chakarov S, Momchilova A, Pankov R. Do we need more human embryonic stem cell lines? Biotechnol Biotechnol Equip. 2010;24:1921–7. doi: 10.2478/V10133-010-0071-X. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

