Cyclic AMP-Independent Control of Twitching Motility in Pseudomonas aeruginosa
- PMID: 28583947
- PMCID: PMC5527385
- DOI: 10.1128/JB.00188-17
Cyclic AMP-Independent Control of Twitching Motility in Pseudomonas aeruginosa
Abstract
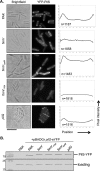
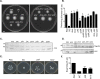
FimV is a Pseudomonas aeruginosa inner membrane hub protein that modulates levels of the second messenger, cyclic AMP (cAMP), through the activation of adenylate cyclase CyaB. Although type IVa pilus (T4aP)-dependent twitching motility is modulated by cAMP levels, mutants lacking FimV are twitching impaired, even when exogenous cAMP is provided. Here we further define FimV's cAMP-dependent and -independent regulation of twitching. We confirmed that the response regulator of the T4aP-associated Chp chemotaxis system, PilG, requires both FimV and the CyaB regulator, FimL, to activate CyaB. However, in cAMP-replete backgrounds-lacking the cAMP phosphodiesterase CpdA or the CheY-like protein PilH or expressing constitutively active CyaB-pilG and fimV mutants failed to twitch. Both cytoplasmic and periplasmic domains of FimV were important for its cAMP-dependent and -independent roles, while its septal peptidoglycan-targeting LysM motif was required only for twitching motility. Polar localization of the sensor kinase PilS, a key regulator of transcription of the major pilin, was FimV dependent. However, unlike its homologues in other species that localize flagellar system components, FimV was not required for swimming motility. These data provide further evidence to support FimV's role as a key hub protein that coordinates the polar localization and function of multiple structural and regulatory proteins involved in P. aeruginosa twitching motility.IMPORTANCEPseudomonas aeruginosa is a serious opportunistic pathogen. Type IVa pili (T4aP) are important for its virulence, because they mediate dissemination and invasion via twitching motility and are involved in surface sensing, which modulates pathogenicity via changes in cAMP levels. Here we show that the hub protein FimV and the response regulator of the Chp system, PilG, regulate twitching independently of their roles in the modulation of cAMP synthesis. These functions do not require the putative scaffold protein FimL, proposed to link PilG with FimV. PilG may regulate asymmetric functioning of the T4aP system to allow for directional movement, while FimV appears to localize both structural and regulatory elements-including the PilSR two-component system-to cell poles for optimal function.
Keywords: Pseudomonas aeruginosa; subcellular localization; twitching motility; type IV pili.
Copyright © 2017 American Society for Microbiology.
Figures



Similar articles
-
The Conserved Tetratricopeptide Repeat-Containing C-Terminal Domain of Pseudomonas aeruginosa FimV Is Required for Its Cyclic AMP-Dependent and -Independent Functions.J Bacteriol. 2016 Jul 28;198(16):2263-74. doi: 10.1128/JB.00322-16. Print 2016 Aug 15. J Bacteriol. 2016. PMID: 27297880 Free PMC article.
-
The peptidoglycan-binding protein FimV promotes assembly of the Pseudomonas aeruginosa type IV pilus secretin.J Bacteriol. 2011 Jan;193(2):540-50. doi: 10.1128/JB.01048-10. Epub 2010 Nov 19. J Bacteriol. 2011. PMID: 21097635 Free PMC article.
-
A scaffold protein connects type IV pili with the Chp chemosensory system to mediate activation of virulence signaling in Pseudomonas aeruginosa.Mol Microbiol. 2016 Aug;101(4):590-605. doi: 10.1111/mmi.13410. Epub 2016 May 27. Mol Microbiol. 2016. PMID: 27145134 Free PMC article.
-
Pseudomonas aeruginosa twitching motility: type IV pili in action.Annu Rev Microbiol. 2012;66:493-520. doi: 10.1146/annurev-micro-092611-150055. Epub 2012 Jul 2. Annu Rev Microbiol. 2012. PMID: 22746331 Review.
-
The molecular genetics of type-4 fimbriae in Pseudomonas aeruginosa--a review.Gene. 1996 Nov 7;179(1):147-55. doi: 10.1016/s0378-1119(96)00441-6. Gene. 1996. PMID: 8955641 Review.
Cited by
-
Surface Sensing for Biofilm Formation in Pseudomonas aeruginosa.Front Microbiol. 2018 Jan 9;8:2671. doi: 10.3389/fmicb.2017.02671. eCollection 2017. Front Microbiol. 2018. PMID: 29375533 Free PMC article. Review.
-
A global genomic approach uncovers novel components for twitching motility-mediated biofilm expansion in Pseudomonas aeruginosa.Microb Genom. 2018 Nov;4(11):e000229. doi: 10.1099/mgen.0.000229. Epub 2018 Nov 1. Microb Genom. 2018. PMID: 30383525 Free PMC article.
-
A bacterial sense of touch: T4P retraction motor as a means of surface sensing by Pseudomonas aeruginosa PA14.J Bacteriol. 2024 Jul 25;206(7):e0044223. doi: 10.1128/jb.00442-23. Epub 2024 Jun 4. J Bacteriol. 2024. PMID: 38832786 Free PMC article. Review.
-
Competitive binding of independent extension and retraction motors explains the quantitative dynamics of type IV pili.Proc Natl Acad Sci U S A. 2021 Feb 23;118(8):e2014926118. doi: 10.1073/pnas.2014926118. Proc Natl Acad Sci U S A. 2021. PMID: 33593905 Free PMC article.
-
Two Acinetobacter baumannii Isolates Obtained From a Fatal Necrotizing Fasciitis Infection Display Distinct Genomic and Phenotypic Characteristics in Comparison to Type Strains.Front Cell Infect Microbiol. 2021 Apr 12;11:635673. doi: 10.3389/fcimb.2021.635673. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 33912474 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

