Ion-pulling simulations provide insights into the mechanisms of channel opening of the skeletal muscle ryanodine receptor
- PMID: 28584051
- PMCID: PMC5546034
- DOI: 10.1074/jbc.M116.760199
Ion-pulling simulations provide insights into the mechanisms of channel opening of the skeletal muscle ryanodine receptor
Abstract
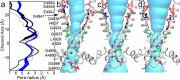
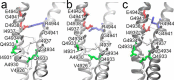
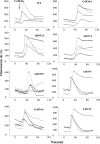
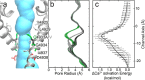
The type 1 ryanodine receptor (RyR1) mediates Ca2+ release from the sarcoplasmic reticulum to initiate skeletal muscle contraction and is associated with muscle diseases, malignant hyperthermia, and central core disease. To better understand RyR1 channel function, we investigated the molecular mechanisms of channel gating and ion permeation. An adequate model of channel gating requires accurate, high-resolution models of both open and closed states of the channel. To this end, we generated an open-channel RyR1 model using molecular simulations to pull Ca2+ through the pore constriction site of a closed-channel RyR1 structure determined at 3.8-Å resolution. Importantly, we find that our open-channel model is consistent with the RyR1 and cardiac RyR (RyR2) open-channel structures reported while this paper was in preparation. Both our model and the published structures show similar rotation of the upper portion of the pore-lining S6 helix away from the 4-fold channel axis and twisting of Ile-4937 at the channel constriction site out of the channel pore. These motions result in a minimum open-channel pore radius of ∼3 Å formed by Gln-4933, rather than Ile-4937 in the closed-channel structure. We also present functional support for our model by mutations around the closed- and open-channel constriction sites (Gln-4933 and Ile-4937). Our results indicate that use of ion-pulling simulations produces a RyR1 open-channel model, which can provide insights into the mechanisms of channel opening complementing those from the structural data.
Keywords: calcium channel; calcium transport; conformational change; molecular dynamics; molecular modeling; ryanodine receptor.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures
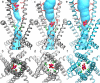
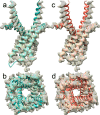
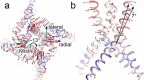

Similar articles
-
G4941K substitution in the pore-lining S6 helix of the skeletal muscle ryanodine receptor increases RyR1 sensitivity to cytosolic and luminal Ca2.J Biol Chem. 2018 Feb 9;293(6):2015-2028. doi: 10.1074/jbc.M117.803247. Epub 2017 Dec 18. J Biol Chem. 2018. PMID: 29255089 Free PMC article.
-
Channel Gating Dependence on Pore Lining Helix Glycine Residues in Skeletal Muscle Ryanodine Receptor.J Biol Chem. 2015 Jul 10;290(28):17535-45. doi: 10.1074/jbc.M115.659672. Epub 2015 May 21. J Biol Chem. 2015. PMID: 25998124 Free PMC article.
-
A central core disease mutation in the Ca2+-binding site of skeletal muscle ryanodine receptor impairs single-channel regulation.Am J Physiol Cell Physiol. 2019 Aug 1;317(2):C358-C365. doi: 10.1152/ajpcell.00052.2019. Epub 2019 Jun 5. Am J Physiol Cell Physiol. 2019. PMID: 31166712 Free PMC article.
-
Role of ryanodine receptors.Crit Rev Biochem Mol Biol. 1994;29(4):229-74. doi: 10.3109/10409239409083482. Crit Rev Biochem Mol Biol. 1994. PMID: 8001396 Review.
-
Ca2+ stores regulate ryanodine receptor Ca2+ release channels via luminal and cytosolic Ca2+ sites.Clin Exp Pharmacol Physiol. 2007 Sep;34(9):889-96. doi: 10.1111/j.1440-1681.2007.04708.x. Clin Exp Pharmacol Physiol. 2007. PMID: 17645636 Review.
Cited by
-
Single-channel properties of skeletal muscle ryanodine receptor pore Δ4923FF4924 in two brothers with a lethal form of fetal akinesia.Cell Calcium. 2020 May;87:102182. doi: 10.1016/j.ceca.2020.102182. Epub 2020 Feb 17. Cell Calcium. 2020. PMID: 32097819 Free PMC article.
-
Molecular Determinants of Mg2+-Mediated Inhibition in RyR1: Insights from Computational Approaches.ACS Omega. 2025 Jul 7;10(28):30757-30772. doi: 10.1021/acsomega.5c03018. eCollection 2025 Jul 22. ACS Omega. 2025. PMID: 40727803 Free PMC article.
-
G4941K substitution in the pore-lining S6 helix of the skeletal muscle ryanodine receptor increases RyR1 sensitivity to cytosolic and luminal Ca2.J Biol Chem. 2018 Feb 9;293(6):2015-2028. doi: 10.1074/jbc.M117.803247. Epub 2017 Dec 18. J Biol Chem. 2018. PMID: 29255089 Free PMC article.
-
Mapping co-regulatory interactions among ligand-binding sites in ryanodine receptor 1.Proteins. 2022 Feb;90(2):385-394. doi: 10.1002/prot.26228. Epub 2021 Sep 6. Proteins. 2022. PMID: 34455637 Free PMC article.
-
In silico assessment of the conduction mechanism of the Ryanodine Receptor 1 reveals previously unknown exit pathways.Sci Rep. 2018 May 2;8(1):6886. doi: 10.1038/s41598-018-25061-z. Sci Rep. 2018. PMID: 29720700 Free PMC article.
References
-
- Franzini-Armstrong C., and Protasi F. (1997) Ryanodine receptors of striated muscles: a complex channel capable of multiple interactions. Physiol. Rev. 77, 699–729 - PubMed
-
- McCarthy T. V., Quane K. A., and Lynch P. J. (2000) Ryanodine receptor mutations in malignant hyperthermia and central core disease. Hum. Mutat. 15, 410–417 - PubMed
-
- Lai F. A., Erickson H. P., Rousseau E., Liu Q. Y., and Meissner G. (1988) Purification and reconstitution of the calcium release channel from skeletal muscle. Nature 331, 315–319 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

