Neurog2 and Ascl1 together regulate a postmitotic derepression circuit to govern laminar fate specification in the murine neocortex
- PMID: 28584103
- PMCID: PMC5488939
- DOI: 10.1073/pnas.1701495114
Neurog2 and Ascl1 together regulate a postmitotic derepression circuit to govern laminar fate specification in the murine neocortex
Abstract
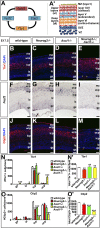
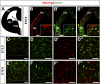
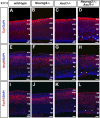
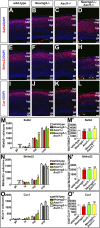
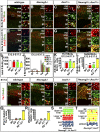
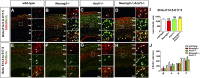
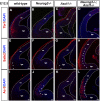
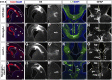
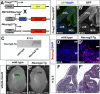
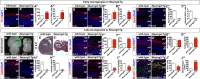
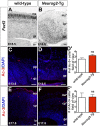
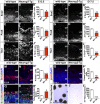
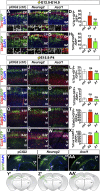
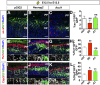
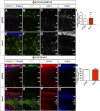
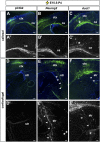
A derepression mode of cell-fate specification involving the transcriptional repressors Tbr1, Fezf2, Satb2, and Ctip2 operates in neocortical projection neurons to specify six layer identities in sequence. Less well understood is how laminar fate transitions are regulated in cortical progenitors. The proneural genes Neurog2 and Ascl1 cooperate in progenitors to control the temporal switch from neurogenesis to gliogenesis. Here we asked whether these proneural genes also regulate laminar fate transitions. Several defects were observed in the derepression circuit in Neurog2-/-;Ascl1-/- mutants: an inability to repress expression of Tbr1 (a deep layer VI marker) during upper-layer neurogenesis, a loss of Fezf2+/Ctip2+ layer V neurons, and precocious differentiation of normally late-born, Satb2+ layer II-IV neurons. Conversely, in stable gain-of-function transgenics, Neurog2 promoted differentiative divisions and extended the period of Tbr1+/Ctip2+ deep-layer neurogenesis while reducing Satb2+ upper-layer neurogenesis. Similarly, acute misexpression of Neurog2 in early cortical progenitors promoted Tbr1 expression, whereas both Neurog2 and Ascl1 induced Ctip2. However, Neurog2 was unable to influence the derepression circuit when misexpressed in late cortical progenitors, and Ascl1 repressed only Satb2. Nevertheless, neurons derived from late misexpression of Neurog2 and, to a lesser extent, Ascl1, extended aberrant subcortical axon projections characteristic of early-born neurons. Finally, Neurog2 and Ascl1 altered the expression of Ikaros and Foxg1, known temporal regulators. Proneural genes thus act in a context-dependent fashion as early determinants, promoting deep-layer neurogenesis in early cortical progenitors via input into the derepression circuit while also influencing other temporal regulators.
Keywords: derepression circuit; laminar fate specification; neocortex; proneural genes; temporal identity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
GSK3 temporally regulates neurogenin 2 proneural activity in the neocortex.J Neurosci. 2012 Jun 6;32(23):7791-805. doi: 10.1523/JNEUROSCI.1309-12.2012. J Neurosci. 2012. PMID: 22674256 Free PMC article.
-
A network of genetic repression and derepression specifies projection fates in the developing neocortex.Proc Natl Acad Sci U S A. 2012 Nov 20;109(47):19071-8. doi: 10.1073/pnas.1216793109. Epub 2012 Nov 9. Proc Natl Acad Sci U S A. 2012. PMID: 23144223 Free PMC article.
-
Neurog2 Acts as a Classical Proneural Gene in the Ventromedial Hypothalamus and Is Required for the Early Phase of Neurogenesis.J Neurosci. 2020 Apr 29;40(18):3549-3563. doi: 10.1523/JNEUROSCI.2610-19.2020. Epub 2020 Apr 9. J Neurosci. 2020. PMID: 32273485 Free PMC article.
-
Instructing neuronal identity during CNS development and astroglial-lineage reprogramming: Roles of NEUROG2 and ASCL1.Brain Res. 2019 Feb 15;1705:66-74. doi: 10.1016/j.brainres.2018.02.045. Epub 2018 Mar 3. Brain Res. 2019. PMID: 29510143 Review.
-
Proneural genes in neocortical development.Neuroscience. 2013 Dec 3;253:256-73. doi: 10.1016/j.neuroscience.2013.08.029. Epub 2013 Aug 30. Neuroscience. 2013. PMID: 23999125 Review.
Cited by
-
Heterogeneity of neurons reprogrammed from spinal cord astrocytes by the proneural factors Ascl1 and Neurogenin2.Cell Rep. 2021 Jul 20;36(3):109409. doi: 10.1016/j.celrep.2021.109409. Cell Rep. 2021. PMID: 34289357 Free PMC article.
-
The Role of Neurodevelopmental Pathways in Brain Tumors.Front Cell Dev Biol. 2021 Apr 27;9:659055. doi: 10.3389/fcell.2021.659055. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34012965 Free PMC article. Review.
-
TAZ Represses the Neuronal Commitment of Neural Stem Cells.Cells. 2020 Oct 2;9(10):2230. doi: 10.3390/cells9102230. Cells. 2020. PMID: 33023162 Free PMC article.
-
Proneural genes define ground-state rules to regulate neurogenic patterning and cortical folding.Neuron. 2021 Sep 15;109(18):2847-2863.e11. doi: 10.1016/j.neuron.2021.07.007. Epub 2021 Aug 17. Neuron. 2021. PMID: 34407390 Free PMC article.
-
A non-canonical role for the proneural gene Neurog1 as a negative regulator of neocortical neurogenesis.Development. 2018 Oct 1;145(19):dev157719. doi: 10.1242/dev.157719. Development. 2018. PMID: 30201687 Free PMC article.
References
-
- Hevner RF, et al. Tbr1 regulates differentiation of the preplate and layer 6. Neuron. 2001;29:353–366. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

