hTERT-Immortalized Bone Mesenchymal Stromal Cells Expressing Rat Galanin via a Single Tetracycline-Inducible Lentivirus System
- PMID: 28584529
- PMCID: PMC5444038
- DOI: 10.1155/2017/6082684
hTERT-Immortalized Bone Mesenchymal Stromal Cells Expressing Rat Galanin via a Single Tetracycline-Inducible Lentivirus System
Abstract
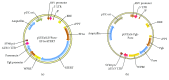
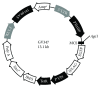
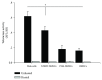
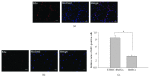
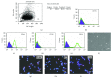
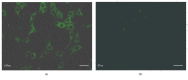
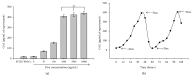
The use of human telomerase reverse transcriptase-immortalized bone marrow mesenchymal stromal cells (hTERT-BMSCs) as vehicles to deliver antinociceptive galanin (GAL) molecules into pain-processing centers represents a novel cell therapy strategy for pain management. Here, an hTERT-BMSCs/Tet-on/GAL cell line was constructed using a single Tet-on-inducible lentivirus system, and subsequent experiments demonstrated that the secretion of rat GAL from hTERT-BMSCs/Tet-on/GAL was switched on and off under the control of an inducer in a dose-dependent manner. The construction of this cell line is the first promising step in the regulation of GAL secretion from hTERT-immortalized BMSCs, and the potential application of this system may provide a stem cell-based research platform for pain.
Figures
References
-
- Dickenson A. H., Chapman V., Green G. M. The pharmacology of excitatory and inhibitory amino acid-mediated events in the transmission and modulation of pain in the spinal cord. General Pharmacology. 1997;28(5):633–638. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

