Modifications of a signal sequence for antibody secretion from insect cells
- PMID: 28584932
- PMCID: PMC6021281
- DOI: 10.1007/s10616-017-0109-0
Modifications of a signal sequence for antibody secretion from insect cells
Abstract
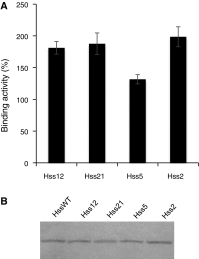
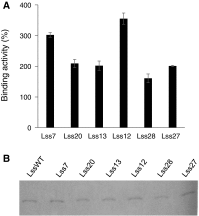
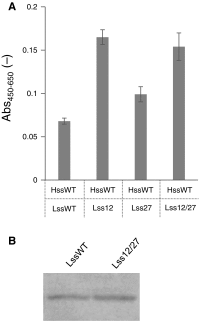
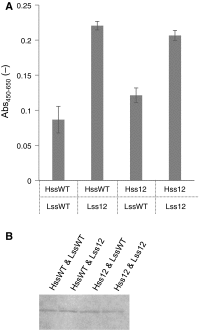
Monoclonal antibodies and antibody fragments have recently been developed for use in diverse diagnostic and therapeutic applications. Insect cells can efficiently secrete recombinant proteins such as antibody molecules through post-translational processing and modifications that are similar to those performed in mammalian cells. In eukaryotic cells, the signal sequence in a nascent polypeptide is recognized by the signal recognition particle, and the polypeptide is then folded and modified in the endoplasmic reticulum. The signal sequence consists of three regions, a positively charged N-terminus, a hydrophobic core, and a polar C-terminus. In the present study, we examined the substitutions of the characteristic amino acids of a Drosophila immunoglobulin heavy chain binding protein signal sequence, and investigated the effect on the secretory production of an antibody Fab fragment from lepidopteran insect cells in transient expression. A modification of the signal sequence for the heavy chain resulted in a twofold increase in the secreted Fab fragment, while the modification for the light chain led to a more than 3.6-fold increase.
Keywords: Antibody; Fab fragment; Insect cell; Recombinant protein production; Secretory production; Signal sequence.
Figures
Similar articles
-
Electrostatic engineering of the interface between heavy and light chains promotes antibody Fab fragment production.Cytotechnology. 2017 Jun;69(3):469-475. doi: 10.1007/s10616-016-9955-4. Epub 2016 Feb 8. Cytotechnology. 2017. PMID: 26856589 Free PMC article.
-
Efficient production of an antibody Fab fragment using the baculovirus-insect cell system.J Biosci Bioeng. 2010 Nov;110(5):577-81. doi: 10.1016/j.jbiosc.2010.06.001. Epub 2010 Jun 29. J Biosci Bioeng. 2010. PMID: 20591732
-
An improved single-chain Fab platform for efficient display and recombinant expression.J Mol Biol. 2015 Jan 30;427(2):576-86. doi: 10.1016/j.jmb.2014.11.017. Epub 2014 Dec 3. J Mol Biol. 2015. PMID: 25481745 Free PMC article.
-
Efficient production of antibody Fab fragment by transient gene expression in insect cells.J Biosci Bioeng. 2017 Aug;124(2):221-226. doi: 10.1016/j.jbiosc.2017.03.007. Epub 2017 Apr 11. J Biosci Bioeng. 2017. PMID: 28410897
-
Pharmacokinetics and biodistribution of genetically-engineered antibodies.Q J Nucl Med. 1998 Dec;42(4):225-41. Q J Nucl Med. 1998. PMID: 9973838 Review.
Cited by
-
FAS2FURIOUS: Moderate-Throughput Secreted Expression of Difficult Recombinant Proteins in Drosophila S2 Cells.Front Bioeng Biotechnol. 2022 May 5;10:871933. doi: 10.3389/fbioe.2022.871933. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35600892 Free PMC article.
-
A comprehensive in silico characterization of bacterial signal peptides for the excretory production of Anabaena variabilis phenylalanine ammonia lyase in Escherichia coli.3 Biotech. 2018 Dec;8(12):488. doi: 10.1007/s13205-018-1517-3. Epub 2018 Nov 16. 3 Biotech. 2018. PMID: 30498661 Free PMC article.
-
ESCargo: a regulatable fluorescent secretory cargo for diverse model organisms.Mol Biol Cell. 2020 Dec 15;31(26):2892-2903. doi: 10.1091/mbc.E20-09-0591. Epub 2020 Oct 28. Mol Biol Cell. 2020. PMID: 33112725 Free PMC article.
-
Essentially Leading Antibody Production: An Investigation of Amino Acids, Myeloma, and Natural V-Region Signal Peptides in Producing Pertuzumab and Trastuzumab Variants.Front Immunol. 2020 Dec 7;11:604318. doi: 10.3389/fimmu.2020.604318. eCollection 2020. Front Immunol. 2020. PMID: 33365032 Free PMC article.
-
Asexual Blood-Stage Malaria Vaccine Candidate PfRipr5: Enhanced Production in Insect Cells.Front Bioeng Biotechnol. 2022 Jun 30;10:908509. doi: 10.3389/fbioe.2022.908509. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35845392 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

