Isolated Mitochondria Transfer Improves Neuronal Differentiation of Schizophrenia-Derived Induced Pluripotent Stem Cells and Rescues Deficits in a Rat Model of the Disorder
- PMID: 28586483
- PMCID: PMC5814822
- DOI: 10.1093/schbul/sbx077
Isolated Mitochondria Transfer Improves Neuronal Differentiation of Schizophrenia-Derived Induced Pluripotent Stem Cells and Rescues Deficits in a Rat Model of the Disorder
Abstract
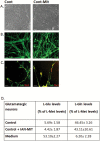
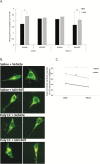
Dysfunction of mitochondria, key players in various essential cell processes, has been repeatedly reported in schizophrenia (SZ). Recently, several studies have reported functional recovery and cellular viability following mitochondrial transplantation, mostly in ischemia experimental models. Here, we aimed to demonstrate beneficial effects of isolated active normal mitochondria (IAN-MIT) transfer in vitro and in vivo, using SZ-derived induced pluripotent stem cells (iPSCs) differentiating into glutamatergic neuron, as well as a rodent model of SZ. First, we show that IAN-MIT enter various cell types without manipulation. Next, we show that IAN-MIT transfer into SZ-derived lymphoblasts induces long-lasting improvement in various mitochondrial functions including cellular oxygen consumption and mitochondrial membrane potential (Δ ψ m). We also demonstrate improved differentiation of SZ-derived iPSCs into neurons, by increased expression of neuronal and glutamatergic markers β3-tubulin, synapsin1, and Tbr1 and by an activation of the glutamate-glutamine cycle. In the animal model, we show that intra-prefrontal cortex injection of IAN-MIT in adolescent rats exposed prenatally to a viral mimic prevents mitochondrial Δ ψ m and attentional deficit at adulthood. Our results provide evidence for a direct link between mitochondrial function and SZ-related deficits both in vitro and in vivo and suggest a therapeutic potential for IAN-MIT transfer in diseases with bioenergetic and neurodevelopmental abnormalities such as SZ.
Figures


References
-
- Ben-Shachar D, Laifenfeld D. Mitochondria, synaptic plasticity, and schizophrenia. Int Rev Neurobiol. 2004;59:273–296. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

