Cell fiber-based three-dimensional culture system for highly efficient expansion of human induced pluripotent stem cells
- PMID: 28588295
- PMCID: PMC5460280
- DOI: 10.1038/s41598-017-03246-2
Cell fiber-based three-dimensional culture system for highly efficient expansion of human induced pluripotent stem cells
Abstract
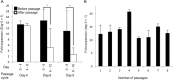
Human pluripotent stem cells are a potentially powerful cellular resource for application in regenerative medicine. Because such applications require large numbers of human pluripotent stem cell-derived cells, a scalable culture system of human pluripotent stem cell needs to be developed. Several suspension culture systems for human pluripotent stem cell expansion exist; however, it is difficult to control the thickness of cell aggregations in these systems, leading to increased cell death likely caused by limited diffusion of gases and nutrients into the aggregations. Here, we describe a scalable culture system using the cell fiber technology for the expansion of human induced pluripotent stem (iPS) cells. The cells were encapsulated and cultured within the core region of core-shell hydrogel microfibers, resulting in the formation of rod-shaped or fiber-shaped cell aggregations with sustained thickness and high viability. By encapsulating the cells with type I collagen, we demonstrated a long-term culture of the cells by serial passaging at a high expansion rate (14-fold in four days) while retaining its pluripotency. Therefore, our culture system could be used for large-scale expansion of human pluripotent stem cells for use in regenerative medicine.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





Similar articles
-
Expansion and long-term maintenance of induced pluripotent stem cells in stirred suspension bioreactors.J Tissue Eng Regen Med. 2012 Jun;6(6):462-72. doi: 10.1002/term.450. Epub 2011 Jul 14. J Tissue Eng Regen Med. 2012. PMID: 21761573
-
Optimizing Human Induced Pluripotent Stem Cell Expansion in Stirred-Suspension Culture.Stem Cells Dev. 2017 Dec 15;26(24):1804-1817. doi: 10.1089/scd.2017.0090. Epub 2017 Nov 21. Stem Cells Dev. 2017. PMID: 29017378
-
Electrospun polystyrene scaffolds as a synthetic substrate for xeno-free expansion and differentiation of human induced pluripotent stem cells.Acta Biomater. 2016 Dec;46:266-277. doi: 10.1016/j.actbio.2016.09.032. Epub 2016 Sep 22. Acta Biomater. 2016. PMID: 27667015
-
Prospects of induced pluripotent stem cell technology in regenerative medicine.Tissue Eng Part B Rev. 2011 Apr;17(2):115-24. doi: 10.1089/ten.TEB.2010.0549. Epub 2011 Feb 15. Tissue Eng Part B Rev. 2011. PMID: 21210760 Review.
-
Scalable Expansion of Pluripotent Stem Cells.Adv Biochem Eng Biotechnol. 2018;163:23-37. doi: 10.1007/10_2017_26. Adv Biochem Eng Biotechnol. 2018. PMID: 29085956 Review.
Cited by
-
Effect of Cyclic Stretch on Tissue Maturation in Myoblast-Laden Hydrogel Fibers.Micromachines (Basel). 2019 Jun 15;10(6):399. doi: 10.3390/mi10060399. Micromachines (Basel). 2019. PMID: 31208059 Free PMC article.
-
Hydrogel-Sheathed hiPSC-Derived Heart Microtissue Enables Anchor-Free Contractile Force Measurement.Adv Sci (Weinh). 2023 Dec;10(35):e2301831. doi: 10.1002/advs.202301831. Epub 2023 Oct 17. Adv Sci (Weinh). 2023. PMID: 37849230 Free PMC article.
-
Microfiber-shaped building-block tissues with endothelial networks for constructing macroscopic tissue assembly.APL Bioeng. 2019 Nov 13;3(4):046101. doi: 10.1063/1.5109966. eCollection 2019 Dec. APL Bioeng. 2019. PMID: 31737859 Free PMC article.
-
Fabrication and Cell Culture Applications of Core-Shell Hydrogel Fibers Composed of Chitosan/DNA Interfacial Polyelectrolyte Complexation and Calcium Alginate: Straight and Beaded Core Variations.Adv Healthc Mater. 2023 Dec;12(31):e2302011. doi: 10.1002/adhm.202302011. Epub 2023 Aug 3. Adv Healthc Mater. 2023. PMID: 37478383 Free PMC article.
-
Glomerulus-on-a-Chip: Current Insights and Future Potential Towards Recapitulating Selectively Permeable Filtration Systems.Int J Nephrol Renovasc Dis. 2022 Mar 10;15:85-101. doi: 10.2147/IJNRD.S344725. eCollection 2022. Int J Nephrol Renovasc Dis. 2022. PMID: 35299832 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

