Transient Hypothyroidism: Dual Effect on Adult-Type Leydig Cell and Sertoli Cell Development
- PMID: 28588502
- PMCID: PMC5441398
- DOI: 10.3389/fphys.2017.00323
Transient Hypothyroidism: Dual Effect on Adult-Type Leydig Cell and Sertoli Cell Development
Abstract
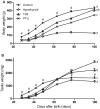
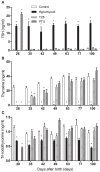
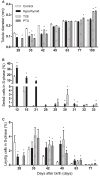
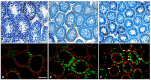
Transient neonatal 6-propyl-2-thiouracil (PTU) induced hypothyroidism affects Leydig and Sertoli cell numbers in the developing testis, resulting in increased adult testis size. The hypothyroid condition was thought to be responsible, an assumption questioned by studies showing that uninterrupted fetal/postnatal hypothyroidism did not affect adult testis size. Here, we investigated effects of transient hypothyroidism on Leydig and Sertoli cell development, employing a perinatal iodide-deficient diet in combination with sodium perchlorate. This hypothyroidism inducing diet was continued until days 1, 7, 14, or 28 postpartum (pp) respectively, when the rats were switched to a euthyroid diet and followed up to adulthood. Continuous euthyroid and hypothyroid, and neonatal PTU-treated rats switched to the euthyroid diet at 28 days pp, were included for comparison. No effects on formation of the adult-type Leydig cell population or on Sertoli cell proliferation and differentiation were observed when the diet switched at/or before day 14 pp. However, when the diet was discontinued at day 28 pp, Leydig cell development was delayed similarly to what was observed in chronic hypothyroid rats. Surprisingly, Sertoli cell proliferation was 6- to 8-fold increased 2 days after the diet switch and remained elevated the next days. In adulthood, Sertoli cell number per seminiferous tubule cross-section and consequently testis weight was increased in this group. These observations implicate that increased adult testis size in transiently hypothyroid rats is not caused by the hypothyroid condition per se, but originates from augmented Sertoli cell proliferation as a consequence of rapid normalization of thyroid hormone concentrations.
Keywords: Leydig cell; Sertoli cell; macroorchidism; proliferation; testis; testosterone; thyroid hormone.
Figures

Similar articles
-
Prenatal induced chronic dietary hypothyroidism delays but does not block adult-type Leydig cell development.Am J Physiol Endocrinol Metab. 2009 Feb;296(2):E305-14. doi: 10.1152/ajpendo.90750.2008. Epub 2008 Nov 25. Am J Physiol Endocrinol Metab. 2009. PMID: 19033542
-
Prolonged and transient neonatal hypothyroidism on Leydig cell differentiation in the postnatal rat testis.Arch Androl. 2004 Sep-Oct;50(5):347-57. doi: 10.1080/01485010490474779. Arch Androl. 2004. PMID: 15551749
-
Impact of neonatal hypothyroidism on Leydig cell number, plasma, and testicular interstitial fluid sex steroids concentration.Endocr Res. 2001 Feb-May;27(1-2):119-41. doi: 10.1081/erc-100107175. Endocr Res. 2001. PMID: 11428705
-
Ontogeny of the androgen receptor expression in the fetal and postnatal testis: its relevance on Sertoli cell maturation and the onset of adult spermatogenesis.Microsc Res Tech. 2009 Nov;72(11):787-95. doi: 10.1002/jemt.20754. Microsc Res Tech. 2009. PMID: 19551717 Review.
-
Thyroid hormones: their role in testicular steroidogenesis.Arch Androl. 2003 Sep-Oct;49(5):375-88. doi: 10.1080/01485010390204968. Arch Androl. 2003. PMID: 12893516 Review.
Cited by
-
Thyroid Hormone Deiodination and Action in the Gonads.Curr Opin Endocr Metab Res. 2018 Oct;2:18-23. doi: 10.1016/j.coemr.2018.01.010. Epub 2018 Feb 7. Curr Opin Endocr Metab Res. 2018. PMID: 30547141 Free PMC article.
-
Roles of thyroid and leptin hormones and their crosstalk in male reproductive functions: an updated review.Endocrine. 2025 Mar;87(3):891-906. doi: 10.1007/s12020-024-04069-8. Epub 2024 Oct 16. Endocrine. 2025. PMID: 39412610 Review.
-
Thyroid dysfunction and semen quality.Int J Immunopathol Pharmacol. 2018 Jan-Dec;32:2058738418775241. doi: 10.1177/2058738418775241. Int J Immunopathol Pharmacol. 2018. PMID: 29737216 Free PMC article. Review.
References
-
- Auharek S. A., De Franca L. R. (2010). Postnatal testis development, Sertoli cell proliferation and number of different spermatogonial types in C57BL/6J mice made transiently hypo- and hyperthyroidic during the neonatal period. J. Anat. 216, 577–588. 10.1111/j.1469-7580.2010.01219.x - DOI - PMC - PubMed
-
- Berndtson W. E., Thompson T. L. (1990). Changing relationships between testis size, Sertoli cell number and spermatogenesis in Sprague-Dawley rats. J. Androl. 11, 429–435. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

