Application of environmental DNA to detect an endangered marine skate species in the wild
- PMID: 28591215
- PMCID: PMC5462358
- DOI: 10.1371/journal.pone.0178124
Application of environmental DNA to detect an endangered marine skate species in the wild
Abstract
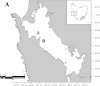
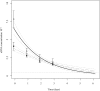
Environmental DNA (eDNA) techniques have only recently been applied in the marine environment to detect the presence of marine species. Species-specific primers and probes were designed to detect the eDNA of the endangered Maugean skate (Zearaja maugeana) from as little as 1 L of water collected at depth (10-15 m) in Macquarie Harbour (MH), Tasmania. The identity of the eDNA was confirmed as Z. maugeana by sequencing the qPCR products and aligning these with the target sequence for a 100% match. This result has validated the use of this eDNA technique for detecting a rare species, Z. maugeana, in the wild. Being able to investigate the presence, and possibly the abundance, of Z. maugeana in MH and Bathurst harbour (BH), would be addressing a conservation imperative for the endangered Z. maugeana. For future application of this technique in the field, the rate of decay was determined for Z. maugeana eDNA under ambient dissolved oxygen (DO) levels (55% saturation) and lower DO (20% saturation) levels, revealing that the eDNA can be detected for 4 and 16 hours respectively, after which eDNA concentration drops below the detection threshold of the assay. With the rate of decay being influenced by starting eDNA concentrations, it is recommended that samples be filtered as soon as possible after collection to minimize further loss of eDNA prior to and during sample processing.
Conflict of interest statement
Figures

Similar articles
-
The endemic and endangered Maugean Skate (Zearaja maugeana) exhibits short-term severe hypoxia tolerance.Conserv Physiol. 2020 Jan 18;8(1):coz105. doi: 10.1093/conphys/coz105. eCollection 2020. Conserv Physiol. 2020. PMID: 31976076 Free PMC article.
-
Development and Validation of Environmental DNA (eDNA) Markers for Detection of Freshwater Turtles.PLoS One. 2015 Jul 22;10(7):e0130965. doi: 10.1371/journal.pone.0130965. eCollection 2015. PLoS One. 2015. PMID: 26200348 Free PMC article.
-
No filters, no fridges: a method for preservation of water samples for eDNA analysis.BMC Res Notes. 2016 Jun 8;9:298. doi: 10.1186/s13104-016-2104-5. BMC Res Notes. 2016. PMID: 27278936 Free PMC article.
-
eDNA as a tool for identifying freshwater species in sustainable forestry: A critical review and potential future applications.Sci Total Environ. 2019 Feb 1;649:1157-1170. doi: 10.1016/j.scitotenv.2018.08.370. Epub 2018 Aug 28. Sci Total Environ. 2019. PMID: 30308887 Review.
-
Current status and topical issues on the use of eDNA-based targeted detection of rare animal species.Sci Total Environ. 2023 Dec 15;904:166675. doi: 10.1016/j.scitotenv.2023.166675. Epub 2023 Aug 28. Sci Total Environ. 2023. PMID: 37647964 Review.
Cited by
-
Metabarcoding reveals unique microbial mat communities and evidence of biogeographic influence in low-oxygen, high-sulfur sinkholes and springs.Ecol Evol. 2024 Mar 24;14(3):e11162. doi: 10.1002/ece3.11162. eCollection 2024 Mar. Ecol Evol. 2024. PMID: 38529029 Free PMC article.
-
A comparison of biomonitoring methodologies for surf zone fish communities.PLoS One. 2023 Jun 14;18(6):e0260903. doi: 10.1371/journal.pone.0260903. eCollection 2023. PLoS One. 2023. PMID: 37314989 Free PMC article.
-
Genome-scale target capture of mitochondrial and nuclear environmental DNA from water samples.Mol Ecol Resour. 2021 Apr;21(3):690-702. doi: 10.1111/1755-0998.13293. Epub 2020 Nov 27. Mol Ecol Resour. 2021. PMID: 33179423 Free PMC article.
-
Application of Omics Tools in Designing and Monitoring Marine Protected Areas For a Sustainable Blue Economy.Front Genet. 2022 Jun 22;13:886494. doi: 10.3389/fgene.2022.886494. eCollection 2022. Front Genet. 2022. PMID: 35812740 Free PMC article.
-
Assessing the efficacy of eDNA metabarcoding for measuring microbial biodiversity within forest ecosystems.Sci Rep. 2021 Jan 15;11(1):1629. doi: 10.1038/s41598-020-80602-9. Sci Rep. 2021. PMID: 33452291 Free PMC article.
References
-
- Goodwin HB. Skate and Ray Management in the Northwest Atlantic: An Overview of Current Management and Recommendations for Conservation [Thesis]. Dalhousie University; 2012.
-
- Sollmann R, Gardner B, Parsons AW, Stocking JJ, Mcclintock BT, Simons TR, et al. A spatial mark-resight model augmented with telemetry data. Ecology. 2013;94(3):553–9. - PubMed
-
- Vaudo JJ, Heithaus MR. Diel and seasonal variation in the use of a nearshore sandflat by a ray community in a near pristine system. Mar Freshw Res. 2012;63(11):1077.
-
- Jerde CL, Mahon AR, Chadderton WL, Lodge DM. “Sight-unseen” detection of rare aquatic species using environmental DNA. Conserv Lett. 2011;4(2):150–7.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

