Electrochemiluminescence Arrays for Studies of Metabolite-related Toxicity
- PMID: 28592918
- PMCID: PMC5459409
- DOI: 10.1002/elan.201600207
Electrochemiluminescence Arrays for Studies of Metabolite-related Toxicity
Abstract
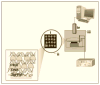
This article reviews recent progress from our laboratory in electrochemiluminescence (ECL) arrays designed for screening toxicity-related chemistry of chemical and drug candidates. Cytochrome P450s and metabolic bioconjugation enzymes convert lipophilic chemicals in our bodies by oxidation and bioconjugation that can lead to toxic metabolites. DNA can be used as an easily measurable toxicity-related endpoint, targeting DNA oxidation and addcut formation with metabolites. ECL using guanosines in the DNA strands as co-reactants have been used in high throughput arrays utilizing DNA-enzyme films fabricated layer-by-layer. This review describes approaches developed to provide new high throughput ECL arrays to aid in toxicity assessment for drug and chemical product development.
Keywords: Cytochrome P450s; DNA Arrays; DNA Damage; Electrochemiluminescence; Metabolites; Toxicity Chemistry.
Figures





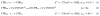





Similar articles
-
Screening Genotoxicity Chemistry with Microfluidic Electrochemiluminescent Arrays.Sensors (Basel). 2017 May 3;17(5):1008. doi: 10.3390/s17051008. Sensors (Basel). 2017. PMID: 28467352 Free PMC article. Review.
-
Modern Approaches to Chemical Toxicity Screening.Curr Opin Electrochem. 2017 Jun;3(1):18-22. doi: 10.1016/j.coelec.2017.03.013. Epub 2017 Apr 3. Curr Opin Electrochem. 2017. PMID: 29250606 Free PMC article.
-
Metabolic toxicity screening using electrochemiluminescence arrays coupled with enzyme-DNA biocolloid reactors and liquid chromatography-mass spectrometry.Annu Rev Anal Chem (Palo Alto Calif). 2012;5(1):79-105. doi: 10.1146/annurev.anchem.111808.073659. Epub 2012 Apr 5. Annu Rev Anal Chem (Palo Alto Calif). 2012. PMID: 22482786 Free PMC article. Review.
-
Biochemical applications of ultrathin films of enzymes, polyions and DNA.Chem Commun (Camb). 2008 Jan 14;(2):141-54. doi: 10.1039/b709121b. Epub 2007 Aug 30. Chem Commun (Camb). 2008. PMID: 18092072 Free PMC article. Review.
-
Elucidating Organ-Specific Metabolic Toxicity Chemistry from Electrochemiluminescent Enzyme/DNA Arrays and Bioreactor Bead-LC-MS/MS.Chem Sci. 2015;6(4):2457-2468. doi: 10.1039/C4SC03401E. Chem Sci. 2015. PMID: 25798217 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
