BPDE-induced genotoxicity: relationship between DNA adducts, mutagenicity in the in vitro PIG-A assay, and the transcriptional response to DNA damage in TK6 cells
- PMID: 28593498
- PMCID: PMC5773665
- DOI: 10.1007/s00204-017-2003-0
BPDE-induced genotoxicity: relationship between DNA adducts, mutagenicity in the in vitro PIG-A assay, and the transcriptional response to DNA damage in TK6 cells
Abstract
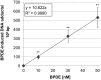
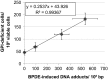
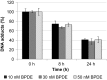
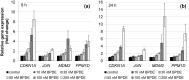
Benzo[a]pyrene is a known human carcinogen. As underlying mechanism, the induction of stable DNA adducts and mutations have been repeatedly demonstrated. Also, the activation of cellular stress response on the transcriptional level has been described. Nevertheless, the interrelationship between these different events is less well understood, especially at low, for human exposure relevant concentrations. Within the present study, we applied the reactive metabolite benzo[a]pyrene diolepoxide (BPDE) in the nanomolar, non-cytotoxic concentration range in human TK6 cells and quantified the induction and repair of stable DNA adducts at the N 2-position of guanine by HPLC with fluorescence detection. Significant levels of DNA lesions were detected even at the lowest concentration of 10 nM BPDE, with a linear increase up to 50 nM. Relative repair was similar at all damage levels, reaching about 30% after 8 h and 60% after 24 h. Mutation frequencies were quantified as GPI-deficient cells by the recently established in vitro PIG-A mutagenicity assay. Again, a linear dose-response-relationship in the before-mentioned concentration range was observed, also when plotting the number of GPI-deficient cells against the number of DNA adducts. Furthermore, we explored the time- and concentration-dependent DNA damage response on the transcriptional level via a high-throughput RT-qPCR technique by quantifying the impact of BPDE on the transcription of 95 genes comprising DNA damage response, DNA repair factors, oxidative stress response, cell cycle arrest, cell proliferation, and apoptosis. As expected, BPDE activated DNA damage signaling, p53 and AP-1 dependent signaling, oxidative stress response, and apoptosis. However, in contrast to DNA adducts and mutations, the onset of the transcriptional DNA damage response was restricted to higher concentrations, indicating that its respective activations require a certain level of DNA lesions. Altogether, the results indicate that in case of BPDE, DNA lesions and mutations were correlated at all concentrations, suggesting that repair is not complete even at low levels of DNA damage. Considering the ongoing discussion on potential thresholds also for genotoxic carcinogens, the results are of major relevance, both with respect to basic research as well as to risk assessment of chemical carcinogens.
Keywords: BPDE; DNA adducts; DNA repair; Gene expression profiling; High throughput RT-qPCR; Mutations.
Conflict of interest statement
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no competing interests.
Figures



References
-
- Cai Y, Patel DJ, Geacintov NE, Broyde S. Dynamics of a benzo[a]pyrene-derived guanine DNA lesion in TGT and CGC sequence contexts: enhanced mobility in TGT explains conformational heterogeneity, flexible bending, and greater susceptibility to nucleotide excision repair. J Mol Biol. 2007;374(2):292–305. doi: 10.1016/j.jmb.2007.09.034. - DOI - PMC - PubMed
-
- Camenisch U, Naegeli H. Role of DNA repair in the protection against genotoxic stress. EXS. 2009;99:111–150. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

