Expanding Axonal Transcriptome Brings New Functions for Axonally Synthesized Proteins in Health and Disease
- PMID: 28593814
- PMCID: PMC5985154
- DOI: 10.1177/1073858417712668
Expanding Axonal Transcriptome Brings New Functions for Axonally Synthesized Proteins in Health and Disease
Abstract
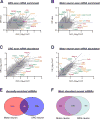
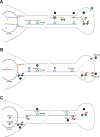
Intra-axonal protein synthesis has been shown to play critical roles in both development and repair of axons. Axons provide long-range connectivity in the nervous system, and disruption of their function and/or structure is seen in several neurological diseases and disorders. Axonally synthesized proteins or losses in axonally synthesized proteins contribute to neurodegenerative diseases, neuropathic pain, viral transport, and survival of axons. Increasing sensitivity of RNA detection and quantitation coupled with methods to isolate axons to purity has shown that a surprisingly complex transcriptome exists in axons. This extends across different species, neuronal populations, and physiological conditions. These studies have helped define the repertoire of neuronal mRNAs that can localize into axons and imply previously unrecognized functions for local translation in neurons. Here, we review the current state of transcriptomics studies of isolated axons, contrast axonal mRNA profiles between different neuronal types and growth states, and discuss how mRNA transport into and translation within axons contribute to neurological disorders.
Keywords: RNA binding protein; axon; mRNA transport; ribonucleoprotein particle; translation.
Figures

Similar articles
-
Axonal mRNA transport and translation at a glance.J Cell Sci. 2018 Apr 13;131(8):jcs196808. doi: 10.1242/jcs.196808. J Cell Sci. 2018. PMID: 29654160 Free PMC article. Review.
-
Molecular determinants of the axonal mRNA transcriptome.Dev Neurobiol. 2014 Mar;74(3):218-32. doi: 10.1002/dneu.22123. Epub 2013 Oct 7. Dev Neurobiol. 2014. PMID: 23959706 Free PMC article. Review.
-
Axonally synthesized β-actin and GAP-43 proteins support distinct modes of axonal growth.J Neurosci. 2013 Feb 20;33(8):3311-22. doi: 10.1523/JNEUROSCI.1722-12.2013. J Neurosci. 2013. PMID: 23426659 Free PMC article.
-
Axonal mRNA localization and translation: local events with broad roles.Cell Mol Life Sci. 2021 Dec;78(23):7379-7395. doi: 10.1007/s00018-021-03995-4. Epub 2021 Oct 26. Cell Mol Life Sci. 2021. PMID: 34698881 Free PMC article. Review.
-
New insights into mRNA trafficking in axons.Dev Neurobiol. 2014 Mar;74(3):233-44. doi: 10.1002/dneu.22121. Epub 2013 Sep 30. Dev Neurobiol. 2014. PMID: 23959656 Review.
Cited by
-
Regulation of Subcellular Protein Synthesis for Restoring Neural Connectivity.Int J Mol Sci. 2025 Jul 28;26(15):7283. doi: 10.3390/ijms26157283. Int J Mol Sci. 2025. PMID: 40806415 Free PMC article. Review.
-
RNA control in pain: Blame it on the messenger.Wiley Interdiscip Rev RNA. 2019 Nov;10(6):e1546. doi: 10.1002/wrna.1546. Epub 2019 May 14. Wiley Interdiscip Rev RNA. 2019. PMID: 31090211 Free PMC article. Review.
-
Protein Translation and Psychiatric Disorders.Neuroscientist. 2020 Feb;26(1):21-42. doi: 10.1177/1073858419853236. Epub 2019 Jul 4. Neuroscientist. 2020. PMID: 31271100 Free PMC article. Review.
-
Age-related loss of axonal regeneration is reflected by the level of local translation.Exp Neurol. 2021 May;339:113594. doi: 10.1016/j.expneurol.2020.113594. Epub 2021 Jan 13. Exp Neurol. 2021. PMID: 33450233 Free PMC article.
-
Axonal mRNA transport and translation at a glance.J Cell Sci. 2018 Apr 13;131(8):jcs196808. doi: 10.1242/jcs.196808. J Cell Sci. 2018. PMID: 29654160 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

