Growth factors for angiogenesis in peripheral arterial disease
- PMID: 28594443
- PMCID: PMC6481523
- DOI: 10.1002/14651858.CD011741.pub2
Growth factors for angiogenesis in peripheral arterial disease
Abstract
Background: Peripheral artery disease (PAD) is associated with a high clinical and socioeconomic burden. Treatments to alleviate the symptoms of PAD and decrease the risks of amputation and death are a high societal priority. A number of growth factors have shown a potential to stimulate angiogenesis. Growth factors delivered directly (as recombinant proteins), or indirectly (e.g. by viral vectors or DNA plasmids encoding these factors), have emerged as a promising strategy to treat patients with PAD.
Objectives: To assess the effects of growth factors that promote angiogenesis for treating people with PAD of the lower extremities.
Search methods: The Cochrane Vascular Information Specialist searched the Specialised Register (June 2016) and CENTRAL (2016, Issue 5). We searched trial registries for details of ongoing or unpublished studies. We also checked the reference lists of relevant publications and, if necessary, tried to contact the trialists for details of the studies.
Selection criteria: We included randomised controlled trials comparing growth factors (delivered directly or indirectly) with no intervention, placebo or any other intervention not based on the growth factor's action in patients with PAD of the lower extremities. The primary outcomes were limb amputation, death and adverse events. The secondary outcomes comprised walking ability, haemodynamic measures, ulceration and rest pain.
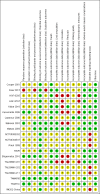
Data collection and analysis: Two review authors independently selected trials and assessed the risk of bias. We used outcomes of the studies at low risk of bias for the main analysis and of all studies in the sensitivity analyses. We calculated odds ratios (OR) for dichotomous outcomes and mean differences for continuous outcomes with 95% confidence intervals (CI). We evaluated statistical heterogeneity using the I2 statistic and Cochrane's Q test. We conducted meta-analysis for the overall effect and for each growth factor as a subgroup analysis using OR in a fixed-effect model. We evaluated the robustness of the results in a sensitivity analysis using risk ratio (RR) and/or a random-effects model. We also assessed the quality of the evidence for each outcome.
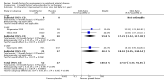
Main results: We included 20 trials in the review and used 14 studies (on approximately 1400 participants) with published results in the analyses. Six published studies compared fibroblast growth factors (FGF), four studies hepatocyte growth factors (HGF) and another four studies vascular endothelial growth factors (VEGF), versus placebo or no therapy. Six of these studies exclusively or mainly investigated participants with intermittent claudication and eight studies exclusively participants with critical limb ischaemia. Follow-up generally ranged from three months to one year. Two small studies provided some data at 2 years and one of them also at 10 years.The direction and size of effects for growth factors on major limb amputations (OR 0.99, 95% CI 0.71 to 1.38; 10 studies, N = 1075) and death (OR 0.99, 95% CI 0.69 to 1.41; 12 studies, N = 1371) at up to two years are uncertain. The quality of the evidence is low due to risk of bias and imprecision (at one year, moderate-quality evidence due to imprecision). However, growth factors may decrease the rate of any limb amputations (OR 0.56, 95% CI 0.31 to 0.99; 6 studies, N = 415). The quality of the evidence is low due to risk of bias and selective reporting.The direction and size of effects for growth factors on serious adverse events (OR 1.09, 95% CI 0.79 to 1.50; 13 studies, N = 1411) and on any adverse events (OR 1.10, 95% CI 0.73 to 1.64; 4 studies, N = 709) at up to two years are also uncertain. The quality of the evidence is low due to risk of bias and imprecision (for serious adverse events at one year, moderate-quality evidence due to imprecision).Growth factors may improve haemodynamic measures (low-quality evidence), ulceration (very low-quality evidence) and rest pain (very low-quality evidence) up to one year, but they have little or no effect on walking ability (low-quality evidence). We did not identify any relevant differences in effects between growth factors (FGF, HGF and VEGF).
Authors' conclusions: The results of this review do not support the use of therapy with the growth factors FGF, HGF or VEGF in people with PAD of the lower extremities to prevent death or major limb amputation or to improve walking ability. However, the use of these growth factors may improve haemodynamic measures and decrease the rate of any limb amputations (probably due to preventing minor amputations) with an uncertain effect on adverse events; an improvement of ulceration and rest pain is very uncertain. New trials at low risk of bias are needed to generate evidence with more certainty.
Conflict of interest statement
VG: institution received funding from the German Federal Ministry of Education and Research (research grant 01KG1411) to conduct the review. MB: institution received funding from the German Federal Ministry of Education and Research (research grant 01KG1411) to conduct the review. AK: institution received funding from the German Federal Ministry of Education and Research (research grant 01KG1411) to conduct the review. AH: institution received funding from the German Federal Ministry of Education and Research (research grant 01KG1411) to conduct the review.
Figures
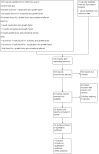

















Update of
- doi: 10.1002/14651858.CD011741
Similar articles
-
Lumbar sympathectomy versus prostanoids for critical limb ischaemia due to non-reconstructable peripheral arterial disease.Cochrane Database Syst Rev. 2018 Apr 16;4(4):CD009366. doi: 10.1002/14651858.CD009366.pub2. Cochrane Database Syst Rev. 2018. PMID: 29658630 Free PMC article.
-
Drugs for preventing postoperative nausea and vomiting in adults after general anaesthesia: a network meta-analysis.Cochrane Database Syst Rev. 2020 Oct 19;10(10):CD012859. doi: 10.1002/14651858.CD012859.pub2. Cochrane Database Syst Rev. 2020. PMID: 33075160 Free PMC article.
-
Exercise for intermittent claudication.Cochrane Database Syst Rev. 2017 Dec 26;12(12):CD000990. doi: 10.1002/14651858.CD000990.pub4. Cochrane Database Syst Rev. 2017. PMID: 29278423 Free PMC article.
-
Effects of additional exercise therapy after a successful vascular intervention for people with symptomatic peripheral arterial disease.Cochrane Database Syst Rev. 2024 May 2;5(5):CD014736. doi: 10.1002/14651858.CD014736.pub2. Cochrane Database Syst Rev. 2024. PMID: 38695785 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
Cited by
-
Use of Growth Factors and Other Cytokines for Treatment of Injuries During a Radiation Public Health Emergency.Radiat Res. 2019 Jul;192(1):99-120. doi: 10.1667/RR15363.1. Epub 2019 May 13. Radiat Res. 2019. PMID: 31081742 Free PMC article.
-
Delivery of hepatocyte growth factor mRNA from nanofibrillar scaffolds in a pig model of peripheral arterial disease.Regen Med. 2020 Jun;15(6):1761-1773. doi: 10.2217/rme-2020-0023. Epub 2020 Aug 10. Regen Med. 2020. PMID: 32772903 Free PMC article.
-
Lower extremity arterial disease in patients with diabetes: a contemporary narrative review.Cardiovasc Diabetol. 2018 Oct 23;17(1):138. doi: 10.1186/s12933-018-0781-1. Cardiovasc Diabetol. 2018. PMID: 30352589 Free PMC article. Review.
-
Loss of m6A demethylase ALKBH5 promotes post-ischemic angiogenesis via post-transcriptional stabilization of WNT5A.Clin Transl Med. 2021 May;11(5):e402. doi: 10.1002/ctm2.402. Clin Transl Med. 2021. PMID: 34047466 Free PMC article.
-
Angiogenesis-related proteins as biomarkers for peripheral artery disease.Heliyon. 2023 Sep 15;9(9):e20166. doi: 10.1016/j.heliyon.2023.e20166. eCollection 2023 Sep. Heliyon. 2023. PMID: 37809892 Free PMC article.
References
References to studies included in this review
Cooper 2001 {published data only}
-
- Cooper LT Jr, Hiatt WR, Creager MA, Regensteiner JG, Casscells, Isner W, et al. Proteinuria in a placebo‐controlled study of basic fibroblast growth factor for intermittent claudication. Vascular Medicine 2001;6(4):235‐9. - PubMed
Deev 2015 {published data only}
-
- Bozo IY, Deev RV, Plaksa IL, Mzhavanadze ND, Chervyakov YV, Staroverov IN, et al. Long‐term results of PCMV‐VEGF165 intramuscular gene transfer in patients with chronic lower limb ischemia. Molecular Therapy 2015;23(Suppl 1):S74. - PubMed
-
- Deev R, Bozo I, Mzhavanadze N, Voronov D, Gavrilenko A, Chervyakov Y, et al. pCMV‐vegf165 intramuscular gene transfer is an effective method of treatment for patients with chronic lower limb ischemia. Journal of Cardiovascular Pharmacology and Therapeutics 2015;20(5):473‐82. - PubMed
HGF‐0205 {published data only}
-
- NCT00189540. Study of hepatocyte growth factor (HGF) via plasmid vector to improve perfusion in critical limb ischemia patients with peripheral ischemic ulcers [A phase II double‐blind, randomized, placebo‐controlled study to assess the safety and efficacy of AMG0001 to improve perfusion in critical limb ischemia in subjects who have peripheral ischemic ulcers]. clinicaltrials.gov/ct2/show/NCT00189540 (accessed 14 July 2015).
-
- Powell RJ, Goodney P, Mendelsohn FO, Moen EK, Annex BH, HGF‐0205 Trial Investigators. Safety and efficacy of patient specific intramuscular injection of HGF plasmid gene therapy on limb perfusion and wound healing in patients with ischemic lower extremity ulceration: results of the HGF‐0205 trial. Journal of Vascular Surgery 2010;52(6):1525‐30. - PMC - PubMed
HGF‐STAT {published data only}
-
- NCT00060892. A Phase II Double‐Blind, Randomized, Placebo‐Controlled Study to Assess the Safety and Efficacy of AMG0001 to Improve Perfusion in Critical Leg Ischemia. clinicaltrials.gov/ct2/show/NCT00060892 (first received 15 May 2003). - PubMed
-
- Powell RJ. Protocol#0207‐546: a phase I/II, double‐blind, randomized, placebo‐controlled study to assess the safety and efficacy of AMG0001 to improve perfusion in critical leg ischemia. Human Gene Therapy 2003; Vol. 14, issue 3:302‐6. [CRS: 8600100000005337] - PubMed
-
- Powell RJ, Dormandy J, Simons M, Morishita R, Annex BH. Therapeutic angiogenesis for critical limb ischemia: design of the hepatocyte growth factor therapeutic angiogenesis clinical trial. Vascular Medicine 2004;9(3):193‐8. - PubMed
-
- Powell RJ, Simons M, Mendelsohn FO, Daniel G, Henry TD, Koga M, et al. Results of a double‐blind, placebo‐controlled study to assess the safety of intramuscular injection of hepatocyte growth factor plasmid to improve limb perfusion in patients with critical limb ischemia. Circulation 2008;118(1):58‐65. - PubMed
Kibbe 2016 {published data only}
-
- Kibbe MR, Hirsch AT, Mendelsohn FO, Davies MG, Pham H, Saucedo J, et al. Safety and efficacy of plasmid DNA expressing two isoforms of hepatocyte growth factor in patients with critical limb ischemia. Gene Therapy 2016;23:306‐12, 399 (corrigendum). - PubMed
-
- NCT01064440. Safety and efficacy study using gene therapy for critical limb ischemia. clinicaltrials.gov/ct2/show/NCT01064440?term=NCT01064440&rank=1 (first received 4 February 2010).
-
- Perin EC, Mendelsohn F, Davies M, Pham H, Saucedo J, Hirsch A, et al. A phase 2, double‐blind, randomized, placebo‐controlled, multicenter trial of the safety and efficacy of plasmid DNA expressing 2 isoforms of hepatocyte growth factor in patients with critical limb ischemia. Journal of the American College of Cardiology 2014;63(12):A2092.
Kusumanto 2006 {published data only}
-
- Kusumanto YH, Mulder NH, Dullaart RPF, Dungen JJAM, Gans ROB. Phase III comparison of intramuscular delivery of ANG1 (a vascular endothelial growth factor containing plasmid) with placebo in diabetic patients with critical limb ischaemia. Molecular Therapy 2001;3:S73.
-
- Kusumanto YH, Weel V, Mulder NH, Smit AJ, Dungen JJ, Hooymans JM, et al. Treatment with intramuscular vascular endothelial growth factor gene compared with placebo for patients with diabetes mellitus and critical limb ischemia: a double‐blind randomized trial. Human Gene Therapy 2006;17(6):683‐91. - PubMed
Lazarous 2000 {published data only}
-
- Lazarous DF, Unger EF, Epstein SE, Stine A, Arevalo JL, Chew EY, et al. Basic fibroblast growth factor in patients with intermittent claudication: results of a phase I trial. Journal of the American College of Cardiology 2000;36(4):1239‐44. - PubMed
Makinen 2002 {published data only}
-
- Makinen K, Manninen H, Hedman M, Matsi P, Mussalo H, Alhava E, et al. Increased vascularity detected by digital subtraction angiography after VEGF gene transfer to human lower limb artery: a randomized, placebo‐controlled, double‐blinded phase II study. Molecular Therapy 2002;6(1):127‐33. - PubMed
-
- Muona K, Mäkinen K, Hedman M, Manninen H, Yla S. 10‐year safety follow‐up in patients with local VEGF gene transfer to ischemic lower limb. Herttuala Gene Therapy 2012;19:392–5. - PubMed
Matyas 2005 {published data only}
-
- Matyas L, Schulte KL, Dormandy JA, Norgren L, Sowade O, Grotzbach G, et al. Arteriogenic gene therapy in patients with unreconstructable critical limb ischemia: a randomized, placebo‐controlled clinical trial of adenovirus 5‐delivered fibroblast growth factor‐4. Human Gene Therapy 2005;16(10):1202‐11. - PubMed
NCT00080392 {published data only}
-
- NCT00080392. EW‐A‐401 to treat intermittent claudication. clinicaltrials.gov/ct2/show/NCT00080392?term=NCT00080392&rank=1 (first received 30 March 2004).
NCT00304837 {published data only}
-
- NCT00304837. VEGF gene transfer for critical limb ischemia. clinicaltrials.gov/ct2/show/NCT00304837?term=NCT00304837&rank=1 (first received 16 March 2006).
Rauh 1999 {published data only}
-
- Rauh G, Gravereaux E, Pieczed A, Curry C, Schainfeld R, Isner JM. Assessment of safety and efficiency of intramuscular gene therapy with VEGF‐2 in patients with critical limb ischaemia. Circulation 1999;100(Suppl 1):770.
RAVE {published data only}
-
- Rajagopalan S, Mohler III E, Lederman RJ, Saucedo J, Mendelsohn FO, Olin J, et al. Regional angiogenesis with vascular endothelial growth factor (VEGF) in peripheral arterial disease: design of the RAVE trial. American Heart Journal 2003;145(6):1114‐8. - PubMed
-
- Rajagopalan S, Mohler III ER, Lederman RJ, Mendelsohn FO, Saucedo JF, Goldman CK, et al. Regional angiogenesis with vascular endothelial growth factor in peripheral arterial disease: a phase II randomized, double‐blind, controlled study of adenoviral delivery of vascular endothelial growth factor 121 in patients with disabling intermittent claudication. Circulation 2003;108(16):1933‐8. - PubMed
Shigematsu 2010 {published data only}
-
- AnGes MG Inc. Academic release of data from phase III clinical trial of HGF gene therapy conducted in Japan. 2007. www.anges‐mg.com/en/news (accessed 4 September 2015).
-
- AnGes MG Inc. Announcement of results of phase III clinical trials of HGF gene therapy in Japan. 2007. www.anges‐mg.com/en/news (accessed 4 September 2015).
-
- Shigematsu H, Yasuda K, Iwai T, Sasajima T, Ishimaru S, Ohashi Y, et al. Randomized, double‐blind, placebo‐controlled clinical trial of hepatocyte growth factor plasmid for critical limb ischemia. Gene Therapy 2010;17(9):1152‐61. - PubMed
-
- Yamada E. Report on long‐term data of collategene (HGF plasmid) ‐ Japanese phase III study for CLI. 2010. www.anges‐mg.com/en/news (accessed 4 September 2015).
TALISMAN‐201 {published data only}
-
- NCT00368797. Efficacy and safety study of NV1FGF in patients with severe peripheral artery occlusive disease (TALISMAN 201). clinicaltrials.gov/ct2/show/NCT00368797?term=NCT00368797&rank=1 (first received 24 August 2006).
-
- Nikol S, Baumgartner I, Belle E, Diehm C, Visona A, Capogrossi MC, et al. Therapeutic angiogenesis with intramuscular NV1FGF improves amputation‐free survival in patients with critical limb ischemia. Molecular Therapy 2008;16(5):972‐8. - PubMed
TALISMAN‐202 {published data only}
-
- Henry TD, Mendelsohn F, Comerota A, Pham E, Grek V, Coleman M, et al. Dose and regimen effects of intramuscular NV1FGF in patients with critical limb ischemia: a randomized, double‐blind, placebo controlled study. European Heart Journal 2006;27(Suppl 1):Abs P1497.
-
- NCT00798005. Efficacy and safety study of NV1FGF in patients with severe peripheral artery occlusive disease (TALISMAN 202). clinicaltrials.gov/ct2/show/NCT00798005?term=NCT00798005&rank=1 (first received 24 November 2008).
TALISMAN‐211 {published data only}
-
- NCT01157871. Dose‐finding, safety and efficacy study of NV1FGF in patients with intermittent claudication (TALISMAN 211). clinicaltrials.gov/ct2/show/NCT01157871?term=NCT01157871&rank=1 (first received 6 July 6 2010).
TAMARIS {published data only}
-
- Belch J, Hiatt WR, Baumgartner I, Driver IV, Nikol S, Norgren L, et al. Effect of fibroblast growth factor NV1FGF on amputation and death: a randomised placebo‐controlled trial of gene therapy in critical limb ischaemia. Lancet 2011;377(9781):1929‐37. - PubMed
-
- NCT00566657. Efficacy and safety of XRP0038/NV1FGF in critical limb ischemia patients with skin lesions (TAMARIS). clinicaltrials.gov/ct2/show/NCT00566657?term=NCT00566657&rank=1 (first received 30 November 2007).
-
- Belle E, Nikol S, Norgren L, Baumgartner I, Driver V, Hiatt W, et al. A randomized, double‐blind placebo‐controlled study of NV1FGF gene therapy in critical limb ischemia patients (TAMARIS Study). Rationale, design and baseline patient characteristics. Archives of Cardiovascular Diseases. Supplements 2011;3:77‐8.
-
- Belle E, Nikol S, Norgren L, Baumgartner I, Driver V, Hiatt WR, et al. Insights on the role of diabetes and geographic variation in patients with critical limb ischaemia. European Journal of Vascular and Endovascular Surgery 2011;42(3):365‐73. - PubMed
TRAFFIC {published data only}
-
- Lederman RJ, Mendelsohn FO, Anderson RD, Saucedo JF, Tenaglia AN, Hermiller JB, et al. Therapeutic angiogenesis with recombinant fibroblast growth factor‐2 for intermittent claudication (the TRAFFIC study): a randomised trial. Lancet 2002;359(9323):2053‐8. - PubMed
-
- Lederman RJ, Tenaglia AN, Anderson RD, Hermiller JB, Rocha‐Singh K, Mendelsohn FO, et al. Design of the therapeutic angiogenesis with recombinant fibroblast growth factor‐2 for intermittent claudication (TRAFFIC) trial. American Journal of Cardiology 2001;88(2):192‐5. - PubMed
VM202‐China {published data only}
-
- NCT01548378. Safety and efficacy study using gene therapy for critical limb ischemia (NL003CLI‐II). clinicaltrials.gov/ct2/show/NCT01548378?term=NCT01548378&rank=1 (first received 5 March 2012).
References to studies excluded from this review
Afshari 2005 {published data only}
-
- Afshari M, Larijani B, Fadayee M, Darvishzadeh F, Ghahary A, Pajouhi M, et al. Efficacy of topical epidermal growth factor in healing diabetic foot ulcers. Therapy 2005;2(5):759‐65.
Anghel 2011 {published data only}
-
- Anghel A, Taranu G, Seclaman E, Rata A, Tamas L, Moldovan H, et al. Safety of vascular endothelial and hepatocyte growth factor gene therapy in patients with critical limb ischemia. Current Neurovascular Research. 2011; Vol. 8:183‐9. [CENTRAL: 811294; CRS: 8600126000002522] - PubMed
Biggs 2009 {published data only}
-
- Biggs T, Dulas D, Duval S, Goldman J, Henry T, Hirsch A, et al. Hepatocyte growth factor gene therapy for patients with critical limb ischemia: Results of a Phase I dose‐escalation trial. Catheterization and Cardiovascular Interventions 2009; Vol. 73, issue S20. [CENTRAL: 793971; CRS: 8600100000008655]
C000000330 {published data only}
-
- C000000330. Peripheral blood mononuclear cell therapy in PAD patients with intermittent claudication randomized trial. upload.umin.ac.jp/cgi‐open‐bin/ctr/ctr.cgi?function=brows&action=bro... (date of disclosure of study information 15 February 2006). [CRS: 8600102000001322]
Capiod 2009 {published data only}
-
- Capiod JC, Tournois C, Vitry F, Sevestre MA, Daliphard S, Reix T, et al. Characterization and comparison of bone marrow and peripheral blood mononuclear cells used for cellular therapy in critical leg ischaemia: towards a new cellular product. Vox Sanguinis 2009; Vol. 96, issue 3:256. [CENTRAL: 747262; CRS: 8600100000008420] - PubMed
Choi 2012 {published data only}
-
- Choi ET, Geraghty P, Cooke C, Schechtman K, Link D, Chambers CM, et al. Stem cell mobilization to treat severe peripheral artery disease (STEMPAD). 2012 Vascular Annual Meeting 2012. [CENTRAL: 831460; CRS: 8600100000018010]
Creager 2011 {published data only}
-
- Creager MA, Olin JW, Belch JJ, Moneta GL, Henry TD, Rajagopalan S, et al. Effect of hypoxia‐inducible factor‐1alpha gene therapy on walking performance in patients with intermittent claudication. Circulation 2011; Vol. 124:1765‐73. [CENTRAL: 805631; CRS: 8600101000001692] - PubMed
Cui 2015 {published data only}
-
- Cui S, Guo L, Li X, Gu Y, Fu J, Dong L, et al. Clinical safety and preliminary efficacy of plasmid pUDK‐HGF expressing human hepatocyte growth factor (HGF) in patients with critical limb ischemia. European Journal of Vascular and Endovascular Surgery 2015;50(4):494‐501. - PubMed
De Leeuw 2008 {published data only}
-
- Leeuw K, Kusumanto Y, Smit AJ, Oomen P, Hoeven JH, Mulder NH, et al. Skin capillary permeability in the diabetic foot with critical limb ischaemia: the effects of a phVEGF165 gene product. Diabetic Medicine 2008; Vol. 25, issue 10:1241‐4. [CRS: 8600100000008110] - PubMed
Domanchuk 2013 {published data only}
Doudar 2013 {published data only}
-
- Doudar N, Ansary M, Abdel Shafy S. Stem cell implantation in the treatment of peripheral vascular disease. Vox Sanguinis 2013; Vol. 105:290. [CENTRAL: 872079; CRS: 8600125000000027]
Fernandez‐Montequín 2009 {published data only}
-
- Fernández‐Montequín JI, Valenzuela‐Silva CM, Díaz OG, Savigne W, Sancho‐Soutelo N, Rivero‐Fernández F, et al. Intra‐lesional injections of recombinant human epidermal growth factor promote granulation and healing in advanced diabetic foot ulcers: multicenter, randomised, placebo‐controlled, double‐blind study. International Wound Journal 2009;6(6):432‐43. - PMC - PubMed
Gavrilenko 2008 {published data only}
-
- Gavrilenko AV, Voronov DA, Konstantinov BA, Bochkov NP. Combination of reconstructive vascular operations with gene‐engineering technologies of angiogenesis stimulation: a present‐day policy aimed at improving the remote results of treating patients with lower limb chronic ischaemia. Angiologiia I Sosudistaia Khirurgiia [Angiology and Vascular Surgery] 2008; Vol. 14, issue 4:49‐53. [CRS: 8600101000001199] - PubMed
Grossman 2007 {published data only}
-
- Grossman PM, Mendelsohn F, Henry TD, Hermiller JB, Litt M, Saucedo, et al. Results from a phase II multicenter, double‐blind placebo‐controlled study of Del‐1 (VLTS‐589) for intermittent claudication in subjects with peripheral arterial disease. American Heart Journal 2007; Vol. 153, issue 5:874. [CRS: 8600100000006669] - PubMed
Huang 2005 {published data only}
-
- Huang P, Li S, Han M, Xiao Z, Yang R, Han ZC. Autologous transplantation of granulocyte colony‐stimulating factor‐mobilized peripheral blood mononuclear cells improves critical limb ischemia in diabetes. Diabetes Care 2005; Vol. 28, issue 9:2155. [CRS: 8600100000006254] - PubMed
Huang 2007 {published data only}
-
- Huang PP, Yang XF, Li SZ, Wen JC, Zhang Y, Han ZC. Randomised comparison of G‐CSF‐mobilized peripheral blood mononuclear cells versus bone marrow‐mononuclear cells for the treatment of patients with lower limb arteriosclerosis obliterans. Thrombosis and Haemostasis 2007; Vol. 98, issue 6:1335‐42. [CRS: 8600100000008050] - PubMed
JPRN‐UMIN000002280 {published data only}
-
- JPRN‐UMIN000002280. Randomized controlled trial of G‐CSF‐mobilized peripheral blood mononuclear cells transplantation for the treatment of patients with peripheral arterial disease. apps.who.int/trialsearch/Trial2.aspx?TrialID=JPRN‐UMIN000002280 (date of first enrolment 08 January 2009). [CENTRAL: 983076; CRS: 8600133000000006]
Kalka 2000 {published data only}
-
- Kalka C, Masuda H, Takahashi T, Gordon R, Tepper O, Gravereaux E, et al. Vascular endothelial growth factor165 gene transfer augments circulating endothelial progenitor cells in human subjects. Circulation Research 2000; Vol. 86, issue 12:1198‐202. [CRS: 8600100000004520] - PubMed
Kirana 2012 {published data only}
-
- Kirana S, Stratmann B, Prante C, Prohaska W, Koerperich H, Lammers D, et al. Autologous stem cell therapy in the treatment of limb ischaemia induced chronic tissue ulcers of diabetic foot patients. International Journal of Clinical Practice 2012; Vol. 66, issue 4:384‐93. [CENTRAL: 830801; CRS: 8600102000000060] - PubMed
Lasala 2012 {published data only}
-
- Lasala GP, Silva JA, Minguell JJ. Therapeutic angiogenesis in patients with severe limb ischemia by transplantation of a combination stem cell product. Journal of Thoracic and Cardiovascular Surgery 2012;144(2):377‐82. - PubMed
-
- Lasala GP, Silva JA, Minguell JJ. Therapeutic angiogenesis in patients with severe limb ischemia by transplantation of an autologous bone marrow‐derived combination stem cell product. Journal of the American College of Cardiology 2011;57(14 Suppl 1):E2020.
Matoba 2008 {published data only}
-
- Matoba S, Tatsumi T, Murohara T, Imaizumi T, Katsuda Y, Ito M, et al. Long‐term clinical outcome after intramuscular implantation of bone marrow mononuclear cells (Therapeutic Angiogenesis by Cell Transplantation [TACT] trial) in patients with chronic limb ischemia. American Heart Journal 2008; Vol. 156, issue 5:1010‐8. [CRS: 8600100000007730] - PubMed
Mohammadzadeh 2013 {published data only}
-
- Mohammadzadeh L, Samedanifard SH, Keshavarzi A, Alimoghaddam K, Larijani B, Ghavamzadeh A, et al. Therapeutic outcomes of transplanting autologous granulocyte colony‐stimulating factor‐mobilised peripheral mononuclear cells in diabetic patients with critical limb ischaemia. Experimental and Clinical Endocrinology & Diabetes 2013; Vol. 121, issue 1:48‐53. [CRS: 8600125000000043] - PubMed
Mohler 2003 {published data only}
-
- Mohler ER III, Rajagopalan S, Olin JW, Trachtenberg JD, Rasmussen H, Pak R, et al. Adenoviral‐mediated gene transfer of vascular endothelial growth factor in critical limb ischemia: safety results from a phase I trial. Vascular Medicine (London) 2003; Vol. 8, issue 1:9‐13. [CRS: 8600100000005500] - PubMed
NCT01041417 {published data only}
-
- NCT01041417. Granulocyte‐macrophage colony stimulating factor (GM‐CSF) and mobilization of progenitor cells in peripheral arterial disease (GPAD‐2). //clinicaltrials.gov/ct2/show/NCT01041417?term=NCT01041417&rank=1 (first received: 29 December 2009).
NCT01302015 {published data only}
-
- NCT01302015. Autologous adipose tissue derived mesenchymal stem cells transplantation in patient with Buerger's disease. clinicaltrials.gov/ct2/show/NCT01302015?term=NCT01302015&rank=1 (first received 17 February 2011). [CENTRAL: 983075; CRS: 8600133000000005]
Niebuhr 2012 {published data only}
-
- Niebuhr A, Henry T, Goldman J, Baumgartner I, Belle E, Gerss J, et al. Long‐term safety of intramuscular gene transfer of non‐viral FGF1 for peripheral artery disease. Gene Therapy 2012;19(3):264‐70. [CRS: 8600126000002424] - PubMed
Olson 2014 {published data only}
-
- Olson E, Demopoulos L, Haws TF, Hu E, Fang Z, Mahar KM, et al. Short‐term treatment with a novel HIF‐prolyl hydroxylase inhibitor (GSK1278863) failed to improve measures of performance in subjects with claudication‐limited peripheral artery disease. Vascular Medicine (London) 2014;19(6):473‐82. - PubMed
Ozturk 2012 {published data only}
-
- Ozturk A, Kucukardali Y, Tangi F, Erikci A, Uzun G, Bashekim C, et al. Therapeutical potential of autologous peripheral blood mononuclear cell transplantation in patients with type 2 diabetic critical limb ischemia. Journal of Diabetes and Its Complications 2012; Vol. 26, issue 1:29‐33. [CENTRAL: 831881; CRS: 8600101000001752] - PubMed
Perin 2011 {published data only}
-
- Perin ECS. A randomized, controlled study of autologous therapy with bone marrow‐derived aldehyde dehydrogenase bright cells in patients with critical limb ischemia. Catheterization and Cardiovascular Interventions 2011; Vol. 78, issue 7:1060‐7. [CRS: 8600101000001591] - PubMed
Poole 2013 {published data only}
Prochazka 2010 {published data only}
Rajagopalan 2002 {published data only}
-
- Rajagopalan S, Trachtenberg J, Mohler E, Olin J, McBride S, Pak R, et al. Phase I study of direct administration of a replication deficient adenovirus vector containing the vascular endothelial growth factor cDNA (CI‐1023) to patients with claudication. American Journal of Cardiology 2002; Vol. 90, issue 5:512‐6. [CRS: 8600100000005299] - PubMed
Senet 2011 {published data only}
-
- Senet P, Vicaut E, Beneton N, Debure C, Lok C, Chosidow O. Topical treatment of hypertensive leg ulcers with platelet‐derived growth factor‐BB: a randomized controlled trial. Archives of Dermatology 2011;147(8):926‐30. - PubMed
Skora 2015 {published data only}
Subramaniyam 2009 {published data only}
-
- Subramaniyam V, Waller EK, Murrow JR, Manatunga A, Lonial S, Kasirajan K, et al. Bone marrow mobilization with granulocyte macrophage colony‐stimulating factor improves endothelial dysfunction and exercise capacity in patients with peripheral arterial disease. American Heart Journal 2009; Vol. 158, issue 1:53. [CRS: 8600101000003485] - PubMed
Tateishi‐Yuyama 2002 {published data only}
-
- Tateishi‐Yuyama E, Matsubara H, Murohara T, Ikeda U, Shintani S, Masaki H, et al. Therapeutic angiogenesis for patients with limb ischaemia by autologous transplantation of bone‐marrow cells: a pilot study and a randomised controlled trial. Lancet 2002; Vol. 360, issue 9331:427. [CRS: 8600100000005197] - PubMed
Van Royen 2005 {published data only}
-
- Royen N, Piek JJ, Legemate DA, Schaper W, Oskam J, Atasever B, et al. Design of the START‐trial: STimulation of ARTeriogenesis using subcutaneous application of GM‐CSF as a new treatment for peripheral vascular disease. A randomized, double‐blind, placebo‐controlled trial. Vascular Medicine 2003;8(3):191‐6. [CRS: 8600100000005615] - PubMed
-
- Royen N, Schirmer SH, Atasever B, Behrens CYH, Ubbink D, Buschmann EE, et al. A pilot study on STimulation of ARTeriogenesis using subcutaneous application of granulocyte‐macrophage colony‐stimulating factor as a new treatment for peripheral vascular disease. Circulation 2005; Vol. 112, issue 7:1040‐6. [CRS: 8600100000006572] - PubMed
Wang 2014 {published data only}
-
- Wang X, Jiang L, Wang X, Yin F, Li G, Feng X, et al. Combination of autologous transplantation of G‐CSF‐mobilized peripheral blood mononuclear cells and panax notoginseng saponins in the treatment of unreconstructable critical limb ischemia. Annals of Vascular Surgery 2014;28(6):1501‐12. - PubMed
Wen 2010 {published data only}
-
- Wen J. Autologous peripheral blood mononuclear cells transplantation in treatment of 30 cases of critical limb ischemia: 3‐year safety follow‐up. Journal of Clinical Rehabilitative Tissue Engineering Research 2010; Vol. 14, issue 45:8526‐30. [CENTRAL: 831687; CRS: 8600101000001544]
Yonemitsu 2013 {published data only}
Zafarghandi 2010 {published data only}
-
- Zafarghandi MRR, Fazel AP, Baharvand H. Safety and efficacy of granulocytecolony‐stimulating factor administration following autologous intramuscular implantation of bone marrow mononuclear cells: a randomized controlled trial in patients with advanced lower limb ischemia. Cytotherapy 2010;12(6):783‐91. [CENTRAL: 781692; CRS: 8600101000001383] - PubMed
References to ongoing studies
AGILITY {published data only}
-
- AnGes MG Inc. AnGes announces amendment to the global development of HGF Plasmid for critical limb ischemia. 2016. www.anges‐mg.com/en/news (accessed 27 June 2016).
-
- AnGes MG Inc. AnGes to start global phase III clinical trials of HGF gene therapy for critical limb ischemia in Europe. 2014. www.anges‐mg.com/en/news (accessed 4 September 2015).
-
- NCT02144610. Efficacy and safety of AMG0001 in subjects with critical limb ischemia (AGILITY). clinicaltrials.gov/ct2/show/NCT02144610?term=NCT02144610&rank=1 (first received 20 May 2014).
NCT00424866 {published data only}
-
- NCT00424866. FGF‐1 for intramuscular injection for the treatment of peripheral arterial disease. clinicaltrials.gov/ct2/show/NCT00424866?term=NCT00424866&rank=1 (first received 18 January 2007).
NCT02276937 {published data only}
-
- NCT02276937. Randomized Phase IIb Trial of DVC1‐0101. clinicaltrials.gov/ct2/show/NCT02276937?term=NCT02276937&rank=1 (first received 22 October 2014).
Additional references
Balshem 2011
-
- Balshem H, Helfand M, Schunemann HJ, Oxman AD, Kunz R, Brozek J, et al. GRADE guidelines: 3. Rating the quality of evidence. Journal of Clinical Epidemiology 2011;64:401‐6. - PubMed
Bedenis 2014
De Backer 2012
De Haro 2009
-
- Haro J, Acin F, Lopez‐Quintana A, Florez A, Martinez‐Aguilar E, Varela C. Meta‐analysis of randomized, controlled clinical trials in angiogenesis: gene and cell therapy in peripheral arterial disease. Heart Vessels 2009;24:321‐8. - PubMed
Fowkes 2013
-
- Fowkes FG, Rudan D, Rudan I, Aboyans V, Denenberg JO, McDermott MM, et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet 2013;382(9901):1329‐40. - PubMed
Gardner 2008
Ghosh 2008
-
- Ghosh R, Walsh SR, Tang TY, Noorani A, Hayes PD. Gene therapy as a novel therapeutic option in the treatment of peripheral vascular disease: systematic review and meta‐analysis. International Journal of Clinical Practice 2008;62(9):1383‐90. - PubMed
Hammer 2013
-
- Hammer A, Steiner S. Gene therapy for therapeutic angiogenesis in peripheral arterial disease ‐ a systematic review and meta‐analysis of randomized, controlled trials. Vasa 2013;42(5):331‐9. - PubMed
Higgins 2011
-
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available at www.cochrane‐handbook.org.
Hirsch 2006
-
- Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL, et al. ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter‐Society Consensus; and Vascular Disease Foundation. Circulation 2006;113:e463‐e654. [DOI: 10.1161/CIRCULATIONAHA.106.174526] - DOI - PubMed
Lane 2014
Norgren 2007
-
- Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FG. Inter‐Society Consensus for the Management of Peripheral Arterial Disease (TASC II). Journal of Vascular Surgery 2007;45 Suppl S:S5A‐S67A. - PubMed
Ouma 2012
Pacilli 2010
-
- Pacilli A, Faggioli G, Stella A, Pasquinelli G. An update on therapeutic angiogenesis for peripheral vascular disease. Annals of Vascular Surgery 2010;24(2):258‐68. - PubMed
Powell 2012
-
- Powell RJ. Update on clinical trials evaluating the effect of biologic therapy in patients with critical limb ischemia. Journal of Vascular Surgery 2012;56(1):264‐6. - PubMed
RevMan 2014 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Rooke 2011
-
- Rooke TW, Hirsch AT, Misra S, Sidawy AN, Beckman JA, Findeiss LK, et al. 2011 ACCF/AHA focused update of the guideline for the management of patients with peripheral artery disease (updating the 2005 guideline): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2011;124(18):2020‐45. - PubMed
Salhiyyah 2015
Tendera 2011
-
- Tendera M, Aboyans V, Bartelink ML, Baumgartner I, Clement D, Collet JP, et al. ESC Guidelines on the diagnosis and treatment of peripheral artery diseases: document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries: the Task Force on the Diagnosis and Treatment of Peripheral Artery Diseases of the European Society of Cardiology (ESC). European Heart Journal 2011;32(22):2851‐906. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

