Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade
- PMID: 28596308
- PMCID: PMC5576142
- DOI: 10.1126/science.aan6733
Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade
Abstract
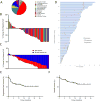
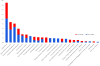
The genomes of cancers deficient in mismatch repair contain exceptionally high numbers of somatic mutations. In a proof-of-concept study, we previously showed that colorectal cancers with mismatch repair deficiency were sensitive to immune checkpoint blockade with antibodies to programmed death receptor-1 (PD-1). We have now expanded this study to evaluate the efficacy of PD-1 blockade in patients with advanced mismatch repair-deficient cancers across 12 different tumor types. Objective radiographic responses were observed in 53% of patients, and complete responses were achieved in 21% of patients. Responses were durable, with median progression-free survival and overall survival still not reached. Functional analysis in a responding patient demonstrated rapid in vivo expansion of neoantigen-specific T cell clones that were reactive to mutant neopeptides found in the tumor. These data support the hypothesis that the large proportion of mutant neoantigens in mismatch repair-deficient cancers make them sensitive to immune checkpoint blockade, regardless of the cancers' tissue of origin.
Copyright © 2017 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
The terms of these arrangements are being managed by Johns Hopkins and Memorial Sloan Kettering in accordance with its conflict of interest policies.
Figures

Comment in
-
In a major shift, cancer drugs go 'tissue-agnostic'.Science. 2017 Jun 16;356(6343):1111-1112. doi: 10.1126/science.356.6343.1111. Science. 2017. PMID: 28619894 No abstract available.
-
Genetic biomarker for cancer immunotherapy.Science. 2017 Jul 28;357(6349):358. doi: 10.1126/science.aao1894. Science. 2017. PMID: 28751597 No abstract available.
-
Biomarkers: Matching mutations to immunotherapy response.Nat Rev Gastroenterol Hepatol. 2017 Oct;14(10):566-567. doi: 10.1038/nrgastro.2017.115. Epub 2017 Aug 16. Nat Rev Gastroenterol Hepatol. 2017. PMID: 28811675 No abstract available.
References
-
- Tumeh PC, Harview CL, Yearley JH, Shintaku IP, Taylor EJ, Robert L, Chmielowski B, Spasic M, Henry G, Ciobanu V, West AN, Carmona M, Kivork C, Seja E, Cherry G, Gutierrez AJ, Grogan TR, Mateus C, Tomasic G, Glaspy JA, Emerson RO, Robins H, Pierce RH, Elashoff DA, Robert C, Ribas A. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature. 2014;515:568–571. doi: 10.1038/nature13954. - DOI - PMC - PubMed
-
- McDermott DF, Drake CG, Sznol M, Choueiri TK, Powderly JD, Smith DC, Brahmer JR, Carvajal RD, Hammers HJ, Puzanov I, Hodi FS, Kluger HM, Topalian SL, Pardoll DM, Wigginton JM, Kollia GD, Gupta A, McDonald D, Sankar V, Sosman JA, Atkins MB. Survival, durable response, and long-term safety in patients with previously treated advanced renal cell carcinoma receiving nivolumab. J Clin Oncol. 2015;33:2013–2020. doi: 10.1200/JCO.2014.58.1041. - DOI - PMC - PubMed
-
- Topalian SL, Sznol M, McDermott DF, Kluger HM, Carvajal RD, Sharfman WH, Brahmer JR, Lawrence DP, Atkins MB, Powderly JD, Leming PD, Lipson EJ, Puzanov I, Smith DC, Taube JM, Wigginton JM, Kollia GD, Gupta A, Pardoll DM, Sosman JA, Hodi FS. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32:1020–1030. doi: 10.1200/JCO.2013.53.0105. - DOI - PMC - PubMed
-
- Gettinger SN, Horn L, Gandhi L, Spigel DR, Antonia SJ, Rizvi NA, Powderly JD, Heist RS, Carvajal RD, Jackman DM, Sequist LV, Smith DC, Leming P, Carbone DP, Pinder-Schenck MC, Topalian SL, Hodi FS, Sosman JA, Sznol M, McDermott DF, Pardoll DM, Sankar V, Ahlers CM, Salvati M, Wigginton JM, Hellmann MD, Kollia GD, Gupta AK, Brahmer JR. Overall survival and long-term safety of nivolumab (anti-programmed death 1 antibody, BMS-936558, ONO-4538) in patients with previously treated advanced non-small-cell lung cancer. J Clin Oncol. 2015;33:2004–2012. doi: 10.1200/JCO.2014.58.3708. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
- U01 CA067941/CA/NCI NIH HHS/United States
- T32 GM007309/GM/NIGMS NIH HHS/United States
- K12 CA090625/CA/NCI NIH HHS/United States
- HHMI/Howard Hughes Medical Institute/United States
- P50 CA098252/CA/NCI NIH HHS/United States
- R01 CA203891/CA/NCI NIH HHS/United States
- R37 CA057345/CA/NCI NIH HHS/United States
- P30 CA016058/CA/NCI NIH HHS/United States
- R01 CA057345/CA/NCI NIH HHS/United States
- K23 CA163672/CA/NCI NIH HHS/United States
- R01 CA067941/CA/NCI NIH HHS/United States
- R37 CA043460/CA/NCI NIH HHS/United States
- P50 CA062924/CA/NCI NIH HHS/United States
- P30 CA006973/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

