Computational analysis of multimorbidity between asthma, eczema and rhinitis
- PMID: 28598986
- PMCID: PMC5466323
- DOI: 10.1371/journal.pone.0179125
Computational analysis of multimorbidity between asthma, eczema and rhinitis
Abstract
Background: The mechanisms explaining the co-existence of asthma, eczema and rhinitis (allergic multimorbidity) are largely unknown. We investigated the mechanisms underlying multimorbidity between three main allergic diseases at a molecular level by identifying the proteins and cellular processes that are common to them.
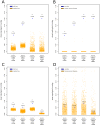
Methods: An in silico study based on computational analysis of the topology of the protein interaction network was performed in order to characterize the molecular mechanisms of multimorbidity of asthma, eczema and rhinitis. As a first step, proteins associated to either disease were identified using data mining approaches, and their overlap was calculated. Secondly, a functional interaction network was built, allowing to identify cellular pathways involved in allergic multimorbidity. Finally, a network-based algorithm generated a ranked list of newly predicted multimorbidity-associated proteins.
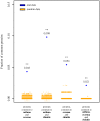
Results: Asthma, eczema and rhinitis shared a larger number of associated proteins than expected by chance, and their associated proteins exhibited a significant degree of interconnectedness in the interaction network. There were 15 pathways involved in the multimorbidity of asthma, eczema and rhinitis, including IL4 signaling and GATA3-related pathways. A number of proteins potentially associated to these multimorbidity processes were also obtained.
Conclusions: These results strongly support the existence of an allergic multimorbidity cluster between asthma, eczema and rhinitis, and suggest that type 2 signaling pathways represent a relevant multimorbidity mechanism of allergic diseases. Furthermore, we identified new candidates contributing to multimorbidity that may assist in identifying new targets for multimorbid allergic diseases.
Conflict of interest statement
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

