Long-term cryopreservation of decellularised oesophagi for tissue engineering clinical application
- PMID: 28599006
- PMCID: PMC5466304
- DOI: 10.1371/journal.pone.0179341
Long-term cryopreservation of decellularised oesophagi for tissue engineering clinical application
Abstract
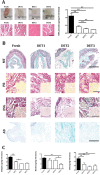
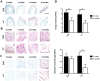
Oesophageal tissue engineering is a therapeutic alternative when oesophageal replacement is required. Decellularised scaffolds are ideal as they are derived from tissue-specific extracellular matrix and are non-immunogenic. However, appropriate preservation may significantly affect scaffold behaviour. Here we aim to prove that an effective method for short- and long-term preservation can be applied to tissue engineered products allowing their translation to clinical application. Rabbit oesophagi were decellularised using the detergent-enzymatic treatment (DET), a combination of deionised water, sodium deoxycholate and DNase-I. Samples were stored in phosphate-buffered saline solution at 4°C (4°C) or slow cooled in medium with 10% Me2SO at -1°C/min followed by storage in liquid nitrogen (SCM). Structural and functional analyses were performed prior to and after 2 and 4 weeks and 3 and 6 months of storage under each condition. Efficient decellularisation was achieved after 2 cycles of DET as determined with histology and DNA quantification, with preservation of the ECM. Only the SCM method, commonly used for cell storage, maintained the architecture and biomechanical properties of the scaffold up to 6 months. On the contrary, 4°C method was effective for short-term storage but led to a progressive distortion and degradation of the tissue architecture at the following time points. Efficient storage allows a timely use of decellularised oesophagi, essential for clinical translation. Here we describe that slow cooling with cryoprotectant solution in liquid nitrogen vapour leads to reliable long-term storage of decellularised oesophageal scaffolds for tissue engineering purposes.
Conflict of interest statement
Figures


References
-
- Mansbridge J. Commercial considerations in tissue engineering. J. Anat. 2006; 209: 527–532. doi: 10.1111/j.1469-7580.2006.00631.x - DOI - PMC - PubMed
-
- Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer. 2010; 127: 2893–2917. doi: 10.1002/ijc.25516 - DOI - PubMed
-
- Spitz L. Oesophageal atresia. Orphanet J. Rare Dis. 2007; 2: 24 doi: 10.1186/1750-1172-2-24 - DOI - PMC - PubMed
-
- Zani A, Pierro A, Elvassore N, De Coppi P. Tissue engineering: an option for esophageal replacement? Semin. Pediatr. Surg. 2009; 18: 57–62. doi: 10.1053/j.sempedsurg.2008.10.011 - DOI - PubMed
-
- Totonelli G, Maghsoudlou P, Garriboli M, Riegler J, Orlando G, Burns AJ, et al. A rat decellularized small bowel scaffold that preserves villus-crypt architecture for intestinal regeneration. Biomaterials. 2012; 33: 3401–3410. doi: 10.1016/j.biomaterials.2012.01.012 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

