HIF-2α promotes the formation of vasculogenic mimicry in pancreatic cancer by regulating the binding of Twist1 to the VE-cadherin promoter
- PMID: 28599281
- PMCID: PMC5564606
- DOI: 10.18632/oncotarget.17999
HIF-2α promotes the formation of vasculogenic mimicry in pancreatic cancer by regulating the binding of Twist1 to the VE-cadherin promoter
Abstract
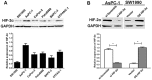
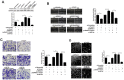
Vasculogenic mimicry (VM) is a blood supply modality that occurs independently of endothelial cell angiogenesis. Hypoxia and the epithelial-mesenchymal transition (EMT) induce VM formation by remodeling the extracellular matrix. Our previous study demonstrated that hypoxia-inducible factor-2 alpha (HIF-2α) promotes the progress of EMT in pancreatic cancer; however, whether HIF-2α promotes VM formation in pancreatic cancer remains unknown. In this study, we investigated HIF-2α expression and VM by immunohistochemistry in 70 pancreatic cancer patients as well as the role of Twist1and Twist2 in HIF-2α-induced VM in vitro and in vivo. We found that the overexpression of HIF-2α and VM were correlated with poor tumor differentiation, late clinical stage and lymph node metastasis, and a poor prognosis in pancreatic cancer. Moreover, the upregulation of HIF-2α in SW1990 cells induced VM formation, whereas the opposite results were found after silencing HIF-2α in AsPC-1 cells. A mechanistic study indicated that HIF-2α might regulate the binding of twist1 to vascular endothelial cadherin (VE-cadherin) to promote VM formation in pancreatic cancer cells, and that the P1 (-421bp) and P4 (-2110bp) regions of the Twist1 binding sequences are positive regulatory elements for VE-cadherin. In addition, we confirmed that the overexpression of HIF-2α increased Twist1 expression and promoted tumor growth and VM formation in pancreatic cancer xenografts in nude mice. These findings indicated that HIF-2α might play a critical role in VM and that HIF-2α and the pathway of HIF-2α inducing VM formation are potential therapeutic targets for pancreatic cancer.
Keywords: HIF-2α; Twist; VE-cadherin; pancreatic cancer; vasculogenic mimicry (VM).
Conflict of interest statement
The authors declare that no potential conflicts of interest exist.
Figures





Similar articles
-
HIF-2α promotes epithelial-mesenchymal transition through regulating Twist2 binding to the promoter of E-cadherin in pancreatic cancer.J Exp Clin Cancer Res. 2016 Feb 3;35:26. doi: 10.1186/s13046-016-0298-y. J Exp Clin Cancer Res. 2016. PMID: 26842802 Free PMC article.
-
Metastasis-associated in colon cancer-1 promotes vasculogenic mimicry in gastric cancer by upregulating TWIST1/2.Oncotarget. 2015 May 10;6(13):11492-506. doi: 10.18632/oncotarget.3416. Oncotarget. 2015. PMID: 25895023 Free PMC article.
-
Hypoxia promotes vasculogenic mimicry formation by inducing epithelial-mesenchymal transition in ovarian carcinoma.Gynecol Oncol. 2014 Jun;133(3):575-83. doi: 10.1016/j.ygyno.2014.02.034. Epub 2014 Feb 28. Gynecol Oncol. 2014. PMID: 24589413
-
Vasculogenic mimicry and tumor metastasis.J BUON. 2016 May-Jun;21(3):533-41. J BUON. 2016. PMID: 27569069 Review.
-
Epithelial-to-endothelial transition and cancer stem cells: two cornerstones of vasculogenic mimicry in malignant tumors.Oncotarget. 2017 May 2;8(18):30502-30510. doi: 10.18632/oncotarget.8461. Oncotarget. 2017. PMID: 27034014 Free PMC article. Review.
Cited by
-
The emerging roles of circular RNAs in vessel co-option and vasculogenic mimicry: clinical insights for anti-angiogenic therapy in cancers.Cancer Metastasis Rev. 2022 Mar;41(1):173-191. doi: 10.1007/s10555-021-10000-8. Epub 2021 Oct 18. Cancer Metastasis Rev. 2022. PMID: 34664157 Review.
-
Domestication and feedback: bidirectional hijacking in pancreatic ductal adenocarcinoma microenvironment.Front Immunol. 2025 Aug 11;16:1585858. doi: 10.3389/fimmu.2025.1585858. eCollection 2025. Front Immunol. 2025. PMID: 40861469 Free PMC article. Review.
-
Resveratrol suppresses serum-induced vasculogenic mimicry through impairing the EphA2/twist-VE-cadherin/AKT pathway in human prostate cancer PC-3 cells.Sci Rep. 2022 Nov 22;12(1):20125. doi: 10.1038/s41598-022-24414-z. Sci Rep. 2022. PMID: 36418859 Free PMC article.
-
Hypoxia signaling in human health and diseases: implications and prospects for therapeutics.Signal Transduct Target Ther. 2022 Jul 7;7(1):218. doi: 10.1038/s41392-022-01080-1. Signal Transduct Target Ther. 2022. PMID: 35798726 Free PMC article. Review.
-
Mechanisms of vasculogenic mimicry in hypoxic tumor microenvironments.Mol Cancer. 2021 Jan 4;20(1):7. doi: 10.1186/s12943-020-01288-1. Mol Cancer. 2021. PMID: 33397409 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

