Neural representation of the self-heard biosonar click in bottlenose dolphins (Tursiops truncatus)
- PMID: 28599518
- PMCID: PMC5438311
- DOI: 10.1121/1.4983191
Neural representation of the self-heard biosonar click in bottlenose dolphins (Tursiops truncatus)
Abstract
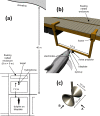
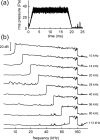
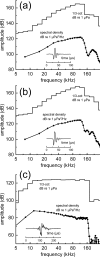
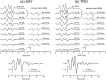
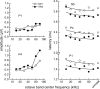
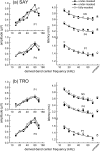
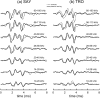
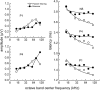
The neural representation of the dolphin broadband biosonar click was investigated by measuring auditory brainstem responses (ABRs) to "self-heard" clicks masked with noise bursts having various high-pass cutoff frequencies. Narrowband ABRs were obtained by sequentially subtracting responses obtained with noise having lower high-pass cutoff frequencies from those obtained with noise having higher cutoff frequencies. For comparison to the biosonar data, ABRs were also measured in a passive listening experiment, where external clicks and masking noise were presented to the dolphins and narrowband ABRs were again derived using the subtractive high-pass noise technique. The results showed little change in the peak latencies of the ABR to the self-heard click from 28 to 113 kHz; i.e., the high-frequency neural responses to the self-heard click were delayed relative to those of an external, spectrally "pink" click. The neural representation of the self-heard click is thus highly synchronous across the echolocation frequencies and does not strongly resemble that of a frequency modulated downsweep (i.e., decreasing-frequency chirp). Longer ABR latencies at higher frequencies are hypothesized to arise from spectral differences between self-heard clicks and external clicks, forward masking from previously emitted biosonar clicks, or neural inhibition accompanying the emission of clicks.
Figures






Similar articles
-
The effects of click rate on the auditory brainstem response of bottlenose dolphins.J Acoust Soc Am. 2017 May;141(5):3396. doi: 10.1121/1.4983447. J Acoust Soc Am. 2017. PMID: 28599539 Free PMC article.
-
Place specificity of the click-evoked auditory brainstem response in the bottlenose dolphin (Tursiops truncatus).J Acoust Soc Am. 2016 Oct;140(4):2593. doi: 10.1121/1.4964274. J Acoust Soc Am. 2016. PMID: 27794308
-
The effects of click and masker spectrum on the auditory brainstem response of bottlenose dolphins (Tursiops truncatus).J Acoust Soc Am. 2016 Oct;140(4):2603. doi: 10.1121/1.4964337. J Acoust Soc Am. 2016. PMID: 27794358
-
Cochlear representation of wideband biosonar sounds and the emergence of neural oscillations.Hear Res. 2025 Jun;462:109261. doi: 10.1016/j.heares.2025.109261. Epub 2025 Apr 5. Hear Res. 2025. PMID: 40286632 Review.
-
Objective neural indices of speech-in-noise perception.Trends Amplif. 2010 Jun;14(2):73-83. doi: 10.1177/1084713810380227. Trends Amplif. 2010. PMID: 20724355 Free PMC article. Review.
Cited by
-
Non-auditory, electrophysiological potentials preceding dolphin biosonar click production.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2018 Mar;204(3):271-283. doi: 10.1007/s00359-017-1234-0. Epub 2017 Dec 8. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2018. PMID: 29222726 Free PMC article.
-
Jittered echo-delay resolution in bottlenose dolphins (Tursiops truncatus).J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2019 Feb;205(1):125-137. doi: 10.1007/s00359-018-1309-6. Epub 2018 Dec 26. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2019. PMID: 30588550 Free PMC article.
References
-
- Au, W. W. L. (1993). The Sonar of Dolphins ( Springer-Verlag, New York: ), 227 pp.
-
- Au, W. W. L. , and Martin, S. W. (2012). “ Why dolphin biosonar performs so well in spite of mediocre ‘equipment,’ ” IET Radar, Sonar Navig. 6, 566−575.10.1049/iet-rsn.2011.0194 - DOI
-
- Au, W. W. L. , and Simmons, J. A. (2007). “ Echolocation in dolphins and bats,” Phys. Today 60(9), 40−45.10.1063/1.2784683 - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

