Functions of intrinsic disorder in transmembrane proteins
- PMID: 28601983
- PMCID: PMC11107515
- DOI: 10.1007/s00018-017-2562-5
Functions of intrinsic disorder in transmembrane proteins
Abstract
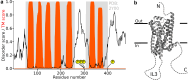
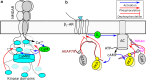
Intrinsic disorder is common in integral membrane proteins, particularly in the intracellular domains. Despite this observation, these domains are not always recognized as being disordered. In this review, we will discuss the biological functions of intrinsically disordered regions of membrane proteins, and address why the flexibility afforded by disorder is mechanistically important. Intrinsically disordered regions are present in many common classes of membrane proteins including ion channels and transporters; G-protein coupled receptors (GPCRs), receptor tyrosine kinases and cytokine receptors. The functions of the disordered regions are many and varied. We will discuss selected examples including: (1) Organization of receptors, kinases, phosphatases and second messenger sources into signaling complexes. (2) Modulation of the membrane-embedded domain function by ball-and-chain like mechanisms. (3) Trafficking of membrane proteins. (4) Transient membrane associations. (5) Post-translational modifications most notably phosphorylation and (6) disorder-linked isoform dependent function. We finish the review by discussing the future challenges facing the membrane protein community regarding protein disorder.
Keywords: Ball-and-chain inhibition; Intrinsically disordered protein; Lipid interaction domain; Membrane protein; Receptor associated signalling complex.
Figures





References
-
- Dunker AK, et al. What’s in a name? Why these proteins are intrinsically disordered. Intrinsically Disord Proteins. 2013;1:1–5. doi: 10.4161/idp.24157. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

