Direct Conversion of Carboxylic Acids to Alkyl Ketones
- PMID: 28604003
- PMCID: PMC5505169
- DOI: 10.1021/acs.orglett.7b01588
Direct Conversion of Carboxylic Acids to Alkyl Ketones
Abstract
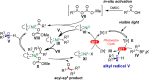
An efficient and mild method for acyl-Csp3 bond formation based on the direct conversion of carboxylic acids has been established. This protocol is enabled by the synergistic, Ir-photoredox/nickel catalytic cross-coupling of in situ activated carboxylic acids and alkyltrifluoroborates. This versatile method is amenable to the cross-coupling of structurally diverse carboxylic acids with various potassium alkyltrifluoroborates, affording the corresponding ketones with high yields. In this operationally simple cross-coupling protocol, aliphatic ketones are obtained in one step from bench stable, readily available carboxylic acids.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Synergistic Photoredox/Nickel Coupling of Acyl Chlorides with Secondary Alkyltrifluoroborates: Dialkyl Ketone Synthesis.J Org Chem. 2017 Feb 3;82(3):1856-1863. doi: 10.1021/acs.joc.6b02897. Epub 2017 Jan 17. J Org Chem. 2017. PMID: 28093913 Free PMC article.
-
Metallaphotoredox-catalysed sp(3)-sp(3) cross-coupling of carboxylic acids with alkyl halides.Nature. 2016 Aug 18;536(7616):322-5. doi: 10.1038/nature19056. Epub 2016 Aug 17. Nature. 2016. PMID: 27535536 Free PMC article.
-
Nickel-Catalyzed Synthesis of Dialkyl Ketones from the Coupling of N-Alkyl Pyridinium Salts with Activated Carboxylic Acids.Angew Chem Int Ed Engl. 2020 Aug 3;59(32):13484-13489. doi: 10.1002/anie.202002271. Epub 2020 Jun 5. Angew Chem Int Ed Engl. 2020. PMID: 32374951 Free PMC article.
-
Carboxylic acids as substrates in homogeneous catalysis.Angew Chem Int Ed Engl. 2008;47(17):3100-20. doi: 10.1002/anie.200704782. Angew Chem Int Ed Engl. 2008. PMID: 18357604 Review.
-
Recent advances in catalytic, enantioselective alpha aminations and alpha oxygenations of carbonyl compounds.Angew Chem Int Ed Engl. 2005 Jul 11;44(28):4292-300. doi: 10.1002/anie.200462314. Angew Chem Int Ed Engl. 2005. PMID: 15945110 Review.
Cited by
-
Asymmetric benzylic C(sp3)-H acylation via dual nickel and photoredox catalysis.Nat Commun. 2021 Jun 10;12(1):3536. doi: 10.1038/s41467-021-23887-2. Nat Commun. 2021. PMID: 34112783 Free PMC article.
-
Ni-Catalyzed Suzuki-Miyaura Cross-Coupling of Aliphatic Amides on the Benchtop.Org Lett. 2020 Jan 3;22(1):1-5. doi: 10.1021/acs.orglett.9b03434. Epub 2019 Oct 17. Org Lett. 2020. PMID: 31621338 Free PMC article.
-
Photoredox Catalysis Enables Access to N-Functionalized 2,1-Borazaronaphthalenes.Org Lett. 2019 Apr 19;21(8):2880-2884. doi: 10.1021/acs.orglett.9b00884. Epub 2019 Mar 27. Org Lett. 2019. PMID: 30916973 Free PMC article.
-
Photochemical Asymmetric Nickel-Catalyzed Acyl Cross-Coupling.Angew Chem Int Ed Engl. 2019 Nov 18;58(47):16854-16858. doi: 10.1002/anie.201910168. Epub 2019 Oct 7. Angew Chem Int Ed Engl. 2019. PMID: 31532568 Free PMC article.
-
Synthesis of C-acyl furanosides via the cross-coupling of glycosyl esters with carboxylic acids.Chem Sci. 2021 Jul 23;12(34):11414-11419. doi: 10.1039/d1sc03596g. eCollection 2021 Sep 1. Chem Sci. 2021. PMID: 34667550 Free PMC article.
References
-
- Huston R. C.; Bailey D. L. J. Am. Chem. Soc. 1946, 68, 1382.10.1021/ja01211a504. - DOI
- House H. O.; Bare T. M. J. Org. Chem. 1968, 33, 943.10.1021/jo01267a003. - DOI
- Suga K.; Watanabe S.; Fujita T.; Takahashi Y. Aust. J. Chem. 1973, 26, 2123.10.1071/CH9732123. - DOI
- Levine R.; Karten M. J.; Kadunce W. M. J. Org. Chem. 1975, 40, 1770.10.1021/jo00900a020. - DOI
-
- Blaise E. E.; Maire M. Compt. Rend. 1907, 145, 73.
- Blaise E. E.; Koehler A. Bull. Soc. Chim. Paris 1910, 7, 215.
- Blaise E. E. Bull. Soc. Chim. 1911, 9, 1.
- Reddy C. K.; Knochel P. Angew. Chem., Int. Ed. Engl. 1996, 35, 1700.10.1002/anie.199617001. - DOI
-
- Hart D. W.; Schwartz J. J. Am. Chem. Soc. 1974, 96, 8115.10.1021/ja00833a048. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

