Recurrent rhinovirus infections in a child with inherited MDA5 deficiency
- PMID: 28606988
- PMCID: PMC5502429
- DOI: 10.1084/jem.20161759
Recurrent rhinovirus infections in a child with inherited MDA5 deficiency
Abstract
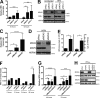
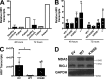
MDA5 is a cytosolic sensor of double-stranded RNA (ds)RNA including viral byproducts and intermediates. We studied a child with life-threatening, recurrent respiratory tract infections, caused by viruses including human rhinovirus (HRV), influenza virus, and respiratory syncytial virus (RSV). We identified in her a homozygous missense mutation in IFIH1 that encodes MDA5. Mutant MDA5 was expressed but did not recognize the synthetic MDA5 agonist/(ds)RNA mimic polyinosinic-polycytidylic acid. When overexpressed, mutant MDA5 failed to drive luciferase activity from the IFNB1 promoter or promoters containing ISRE or NF-κB sequence motifs. In respiratory epithelial cells or fibroblasts, wild-type but not knockdown of MDA5 restricted HRV infection while increasing IFN-stimulated gene expression and IFN-β/λ. However, wild-type MDA5 did not restrict influenza virus or RSV replication. Moreover, nasal epithelial cells from the patient, or fibroblasts gene-edited to express mutant MDA5, showed increased replication of HRV but not influenza or RSV. Thus, human MDA5 deficiency is a novel inborn error of innate and/or intrinsic immunity that causes impaired (ds)RNA sensing, reduced IFN induction, and susceptibility to the common cold virus.
This is a work of the U.S. Government and is not subject to copyright protection in the United States. Foreign copyrights may apply.
Figures








References
-
- Balish A.L., Katz J.M., and Klimov A.I.. 2013. Influenza: propagation, quantification, and storage. Curr Protoc Microbiol Chapter 15:Unit 15G 11. - PubMed
-
- Benitez A.A., Panis M., Xue J., Varble A., Shim J.V., Frick A.L., López C.B., Sachs D., and tenOever B.R.. 2015. In Vivo RNAi Screening Identifies MDA5 as a Significant Contributor to the Cellular Defense against Influenza A Virus. Cell Reports. 11:1714–1726. 10.1016/j.celrep.2015.05.032 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

