Tweety-Homolog 1 Drives Brain Colonization of Gliomas
- PMID: 28607172
- PMCID: PMC6705725
- DOI: 10.1523/JNEUROSCI.3532-16.2017
Tweety-Homolog 1 Drives Brain Colonization of Gliomas
Abstract
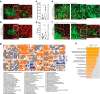
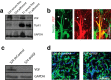
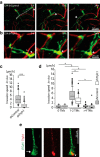
Early and progressive colonization of the healthy brain is one hallmark of diffuse gliomas, including glioblastomas. We recently discovered ultralong (>10 to hundreds of microns) membrane protrusions [tumor microtubes (TMs)] extended by glioma cells. TMs have been associated with the capacity of glioma cells to effectively invade the brain and proliferate. Moreover, TMs are also used by some tumor cells to interconnect to one large, resistant multicellular network. Here, we performed a correlative gene-expression microarray and in vivo imaging analysis, and identified novel molecular candidates for TM formation and function. Interestingly, these genes were previously linked to normal CNS development. One of the genes scoring highest in tests related to the outgrowth of TMs was tweety-homolog 1 (TTYH1), which was highly expressed in a fraction of TMs in mice and patients. Ttyh1 was confirmed to be a potent regulator of normal TM morphology and of TM-mediated tumor-cell invasion and proliferation. Glioma cells with one or two TMs were mainly responsible for effective brain colonization, and Ttyh1 downregulation particularly affected this cellular subtype, resulting in reduced tumor progression and prolonged survival of mice. The remaining Ttyh1-deficient tumor cells, however, had more interconnecting TMs, which were associated with increased radioresistance in those small tumors. These findings imply a cellular and molecular heterogeneity in gliomas regarding formation and function of distinct TM subtypes, with multiple parallels to neuronal development, and suggest that Ttyh1 might be a promising target to specifically reduce TM-associated brain colonization by glioma cells in patients.SIGNIFICANCE STATEMENT In this report, we identify tweety-homolog 1 (Ttyh1), a membrane protein linked to neuronal development, as a potent driver of tumor microtube (TM)-mediated brain colonization by glioma cells. Targeting of Ttyh1 effectively inhibited the formation of invasive TMs and glioma growth, but increased network formation by intercellular TMs, suggesting a functional and molecular heterogeneity of the recently discovered TMs with potential implications for future TM-targeting strategies.
Keywords: Ttyh1; glioblastoma; glioma; invasion; migration; tumor microtubes.
Copyright © 2017 the authors 0270-6474/17/376837-14$15.00/0.
Figures

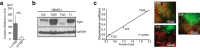




Similar articles
-
Tumor microtubes convey resistance to surgical lesions and chemotherapy in gliomas.Neuro Oncol. 2017 Oct 1;19(10):1316-1326. doi: 10.1093/neuonc/nox070. Neuro Oncol. 2017. PMID: 28419303 Free PMC article.
-
Individual glioblastoma cells harbor both proliferative and invasive capabilities during tumor progression.Neuro Oncol. 2023 Dec 8;25(12):2150-2162. doi: 10.1093/neuonc/noad109. Neuro Oncol. 2023. PMID: 37335907 Free PMC article.
-
Tumor microtubes: A new potential therapeutic target for high-grade gliomas.J Neuropathol Exp Neurol. 2025 Feb 1;84(2):93-103. doi: 10.1093/jnen/nlae119. J Neuropathol Exp Neurol. 2025. PMID: 39560360 Review.
-
A malignant cellular network in gliomas: potential clinical implications.Neuro Oncol. 2016 Apr;18(4):479-85. doi: 10.1093/neuonc/now014. Neuro Oncol. 2016. PMID: 26995789 Free PMC article. Review.
-
Brain tumour cells interconnect to a functional and resistant network.Nature. 2015 Dec 3;528(7580):93-8. doi: 10.1038/nature16071. Epub 2015 Nov 4. Nature. 2015. PMID: 26536111
Cited by
-
Tunneling nanotubes: A bridge for heterogeneity in glioblastoma and a new therapeutic target?Cancer Rep (Hoboken). 2019 Dec;2(6):e1185. doi: 10.1002/cnr2.1185. Epub 2019 May 8. Cancer Rep (Hoboken). 2019. PMID: 32729189 Free PMC article. Review.
-
Pre-Clinical Drug Testing in 2D and 3D Human In Vitro Models of Glioblastoma Incorporating Non-Neoplastic Astrocytes: Tunneling Nano Tubules and Mitochondrial Transfer Modulates Cell Behavior and Therapeutic Respons.Int J Mol Sci. 2019 Nov 29;20(23):6017. doi: 10.3390/ijms20236017. Int J Mol Sci. 2019. PMID: 31795330 Free PMC article.
-
Patient-Derived Tumor Organoids for Guidance of Personalized Drug Therapies in Recurrent Glioblastoma.Int J Mol Sci. 2022 Jun 12;23(12):6572. doi: 10.3390/ijms23126572. Int J Mol Sci. 2022. PMID: 35743016 Free PMC article.
-
TTYH1 and TTYH2 Serve as LRRC8A-Independent Volume-Regulated Anion Channels in Cancer Cells.Cells. 2019 Jun 9;8(6):562. doi: 10.3390/cells8060562. Cells. 2019. PMID: 31181821 Free PMC article.
-
The tweety Gene Family: From Embryo to Disease.Front Mol Neurosci. 2021 Jun 28;14:672511. doi: 10.3389/fnmol.2021.672511. eCollection 2021. Front Mol Neurosci. 2021. PMID: 34262434 Free PMC article.
References
-
- Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate—a practical and powerful approach to multiple testing. J Roy Stat Soc B Met 57:289–300.
-
- Cairncross JG, Wang M, Jenkins RB, Shaw EG, Giannini C, Brachman DG, Buckner JC, Fink KL, Souhami L, Laperriere NJ, Huse JT, Mehta MP, Curran WJ Jr (2014) Benefit from procarbazine, lomustine, and vincristine in oligodendroglial tumors is associated with mutation of IDH. J Clin Oncol 32:783–790. 10.1200/JCO.2013.49.3726 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
