Heteromers of μ opioid and dopamine D1 receptors modulate opioid-induced locomotor sensitization in a dopamine-independent manner
- PMID: 28608532
- PMCID: PMC5554314
- DOI: 10.1111/bph.13908
Heteromers of μ opioid and dopamine D1 receptors modulate opioid-induced locomotor sensitization in a dopamine-independent manner
Abstract
Background and purpose: Exposure to opiates induces locomotor sensitization in rodents, which has been proposed to correspond to the compulsive drug-seeking behaviour. Numerous studies have demonstrated that locomotor sensitization can occur in a dopamine transmission-independent manner; however, the underlying mechanisms are unclear.
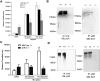
Experimental approach: Co-immunoprecipitation, BRET and cross-antagonism assays were used to demonstrate the existence of receptor heterodimers. Function of heterodimers was evaluated by behavioural studies of locomotor sensitization.
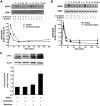
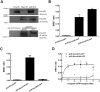
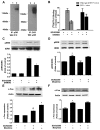
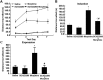
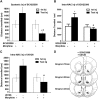
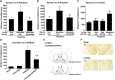
Key results: The dopamine D1 receptor antagonist SCH23390 antagonized the signalling initiated by stimulation of μ opioid receptors with agonists in transfected cells expressing two receptors and in striatal tissues from wild-type but not D1 receptor knockout (KO) mice, suggesting that SCH23390 modified μ receptor function via receptor heteromers, as the ability of an antagonist of one of the receptors to inhibit signals originated by stimulation of the partner receptor was a characteristic of receptor heteromers. The existence of μ receptor-D1 receptor heterodimers was further supported by biochemical and biophysical assays. In vivo, when dopamine release was absent (by destruction of the dopaminergic projection from the ventral tegmental area to the striatum), SCH23390 still significantly inhibited μ receptor agonist-induced behavioural responses in rats. Additionally, we demonstrated that D1 or μ receptor KO mice and thus unable to form μ receptor-D1 receptor heterodimers, failed to show locomotor sensitization to morphine.
Conclusion and implications: Our results suggest that μ receptor-D1 receptor heterodimers may be involved in the dopamine-independent expression of locomotor sensitization to opiates.
© 2017 The British Pharmacological Society.
Figures

Similar articles
-
Systemic morphine-induced Fos protein in the rat striatum and nucleus accumbens is regulated by mu opioid receptors in the substantia nigra and ventral tegmental area.J Neurosci. 1997 Nov 1;17(21):8596-612. doi: 10.1523/JNEUROSCI.17-21-08596.1997. J Neurosci. 1997. PMID: 9334431 Free PMC article.
-
Increased dopamine D2 receptor binding and enhanced apomorphine-induced locomotor activity in mu-opioid receptor knockout mice.Brain Res Bull. 2003 Jun 30;61(1):109-15. doi: 10.1016/s0361-9230(03)00077-7. Brain Res Bull. 2003. PMID: 12788214
-
Preprodynorphin mediates locomotion and D2 dopamine and mu-opioid receptor changes induced by chronic 'binge' cocaine administration.J Neurochem. 2007 Sep;102(6):1817-1830. doi: 10.1111/j.1471-4159.2007.04661.x. Epub 2007 May 26. J Neurochem. 2007. PMID: 17532787
-
A behavioral genetics approach to understanding D1 receptor involvement in phasic dopamine signaling.Mol Cell Neurosci. 2011 Jan;46(1):21-31. doi: 10.1016/j.mcn.2010.09.011. Epub 2010 Oct 1. Mol Cell Neurosci. 2011. PMID: 20888914 Free PMC article. Review.
-
Dopamine receptor interacting proteins (DRIPs) of dopamine D1-like receptors in the central nervous system.Mol Cells. 2008 Apr 30;25(2):149-57. Epub 2008 Mar 28. Mol Cells. 2008. PMID: 18414018 Review.
Cited by
-
Mu Opioid Receptor Heterodimers Emerge as Novel Therapeutic Targets: Recent Progress and Future Perspective.Front Pharmacol. 2020 Jul 15;11:1078. doi: 10.3389/fphar.2020.01078. eCollection 2020. Front Pharmacol. 2020. PMID: 32760281 Free PMC article. Review.
-
The classical D1 dopamine receptor antagonist SCH23390 is a functional sigma-1 receptor allosteric modulator.Acta Pharmacol Sin. 2024 Aug;45(8):1582-1590. doi: 10.1038/s41401-024-01256-1. Epub 2024 Apr 11. Acta Pharmacol Sin. 2024. PMID: 38605179 Free PMC article.
-
Co-localization of mu-opioid and dopamine D1 receptors in the medial preoptic area and bed nucleus of the stria terminalis across seasonal states in male European starlings.Horm Behav. 2019 Jan;107:1-10. doi: 10.1016/j.yhbeh.2018.11.003. Epub 2018 Nov 20. Horm Behav. 2019. PMID: 30423316 Free PMC article.
-
Molecular Imaging of Opioid and Dopamine Systems: Insights Into the Pharmacogenetics of Opioid Use Disorders.Front Psychiatry. 2019 Sep 18;10:626. doi: 10.3389/fpsyt.2019.00626. eCollection 2019. Front Psychiatry. 2019. PMID: 31620026 Free PMC article. Review.
-
Dietary supplementation and the role of phytochemicals against the Alzheimer's disease: Focus on polyphenolic compounds.J Prev Alzheimers Dis. 2025 Jan;12(1):100004. doi: 10.1016/j.tjpad.2024.100004. Epub 2025 Jan 1. J Prev Alzheimers Dis. 2025. PMID: 39800464 Free PMC article. Review.
References
-
- Amalric M, Koob GF (1985). Low doses of methylnaloxonium in the nucleus accumbens antagonize hyperactivity induced by heroin in the rat. Pharmacol Biochem Behav 23: 411–415. - PubMed
-
- Becker A, Grecksch G, Kraus J, Peters B, Schroeder H, Schulz S et al. (2001). Loss of locomotor sensitisation in response to morphine in D1 receptor deficient mice. Naunyn Schmiedebergs Arch Pharmacol 363: 562–568. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

