Kupffer Cell-Derived Tnf Triggers Cholangiocellular Tumorigenesis through JNK due to Chronic Mitochondrial Dysfunction and ROS
- PMID: 28609656
- PMCID: PMC7909318
- DOI: 10.1016/j.ccell.2017.05.006
Kupffer Cell-Derived Tnf Triggers Cholangiocellular Tumorigenesis through JNK due to Chronic Mitochondrial Dysfunction and ROS
Abstract
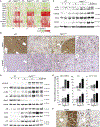
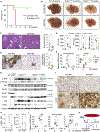
Intrahepatic cholangiocarcinoma (ICC) is a highly malignant, heterogeneous cancer with poor treatment options. We found that mitochondrial dysfunction and oxidative stress trigger a niche favoring cholangiocellular overgrowth and tumorigenesis. Liver damage, reactive oxygen species (ROS) and paracrine tumor necrosis factor (Tnf) from Kupffer cells caused JNK-mediated cholangiocellular proliferation and oncogenic transformation. Anti-oxidant treatment, Kupffer cell depletion, Tnfr1 deletion, or JNK inhibition reduced cholangiocellular pre-neoplastic lesions. Liver-specific JNK1/2 deletion led to tumor reduction and enhanced survival in Akt/Notch- or p53/Kras-induced ICC models. In human ICC, high Tnf expression near ICC lesions, cholangiocellular JNK-phosphorylation, and ROS accumulation in surrounding hepatocytes are present. Thus, Kupffer cell-derived Tnf favors cholangiocellular proliferation/differentiation and carcinogenesis. Targeting the ROS/Tnf/JNK axis may provide opportunities for ICC therapy.
Keywords: JNK; Kupffer cell; Tnf; cholastasis; intrahepatic cholangiocarcinoma; mitochondrial dysfunction; pro-inflammatory niche; reactive oxygen species; unfolded protein response.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures






References
-
- Ariizumi SI, and Yamamoto M (2014). Intrahepatic cholangiocarcinoma and cholangiolocellular carcinoma in cirrhosis and chronic viral hepatitis. Surg. Today 45, 682–687. - PubMed
-
- Arnott CH, Scott KA, Moore RJ, Robinson SC, Thompson RG, and Balkwill FR (2004). Expression of both TNF-alpha receptor subtypes is essential for optimal skin tumour development. Oncogene 23, 1902–1910. - PubMed
-
- Berger E, Yuan D, Waldschmitt N, Rath E, Allgäuer M, Staszewski O, Kober O, Lobner E, Schöttl T, Prinz M, et al. (2016). Mitochondrial function controls epithelial stemness and proliferation in the intestine independent of unfolded protein response signaling mechanisms. Nat. Commun 7, 13171. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

