Cell migration on material-driven fibronectin microenvironments
- PMID: 28612879
- PMCID: PMC5858633
- DOI: 10.1039/c7bm00333a
Cell migration on material-driven fibronectin microenvironments
Abstract
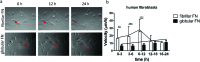
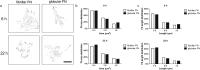
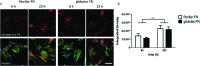
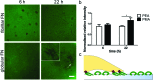
Cell migration is a fundamental process involved in a wide range of biological phenomena. However, how the underlying mechanisms that control migration are orchestrated is not fully understood. In this work, we explore the migratory characteristics of human fibroblasts using different organisations of fibronectin (FN) triggered by two chemically similar surfaces, poly(ethyl acrylate) (PEA) and poly(methyl acrylate) (PMA); cell migration is mediated via an intermediate layer of fibronectin (FN). FN is organised into nanonetworks upon simple adsorption on PEA whereas a globular conformation is observed on PMA. We studied cell speed over the course of 24 h and the morphology of focal adhesions in terms of area and length. Additionally, we analysed the amount of cell-secreted FN as well as FN remodelling. Velocity of human fibroblasts was found to exhibit a biphasic behaviour on PEA, whereas it remained fairly constant on PMA. FA analysis revealed more mature focal adhesions on PEA over time contrary to smaller FAs found on PMA. Finally, human fibroblasts seemed to remodel adsorbed FN more on PMA than on PEA. Overall, these results indicate that the cell-protein-material interface affects cell migratory behaviour. Analysis of FAs together with FN secretion and remodelling were associated with differences in cell velocity providing insights into the factors that can modulate cell motility.
Figures

References
-
- Vicente-Manzanares M., Webb D. J., Horwitz A. R. J. Cell Sci. 2005;118:4917–4919. - PubMed
-
- Lock J. G., Wehrle-Haller B., Strömblad S. Semin. Cancer Biol. 2008;18:65–76. - PubMed
-
- Yamada K. M., Geiger B. Curr. Opin. Cell Biol. 1997;9:76–85. - PubMed
-
- Wozniak M A., Modzelewska K., Kwong L., Keely P. J. Biochim. Biophys. Acta, Mol. Cell Res. 2004;1692:103–119. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

