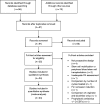
Meta-analysis and indirect treatment comparison of lipegfilgrastim with pegfilgrastim and filgrastim for the reduction of chemotherapy-induced neutropenia-related events
- PMID: 28614980
- PMCID: PMC6094503
- DOI: 10.1177/1078155217714859
Meta-analysis and indirect treatment comparison of lipegfilgrastim with pegfilgrastim and filgrastim for the reduction of chemotherapy-induced neutropenia-related events
Abstract
Background Granulocyte colony-stimulating factors are effective at reducing the risk and duration of neutropenia. The current meta-analysis compared the neutropenia-related efficacy and safety of lipegfilgrastim to those of pegfilgrastim and filgrastim. Methods Embase was searched for trials examining the efficacy/safety of lipegfilgrastim, pegfilgrastim, or filgrastim. Outcomes included febrile neutropenia, severe neutropenia, duration of severe neutropenia, time to recovery of absolute neutrophil count, and incidence of bone pain. Direct comparisons were made using random-effects models. No trials directly compared lipegfilgrastim and filgrastim. Indirect comparisons were made between lipegfilgrastim and filgrastim with pegfilgrastim as the common comparator. Results This meta-analysis included a total of 5769 patients from 24 studies. Over all cycles, lipegfilgrastim showed a lower, nonsignificant risk of febrile neutropenia compared with pegfilgrastim. Lipegfilgrastim has a lower risk of febrile neutropenia versus filgrastim but was also not statistically significant. The risk ratio for severe neutropenia in cycle 1 was 0.80, a 20% reduction in favor of lipegfilgrastim. For cycles 2-4, the risk ratio was 0.53 (0.35, 0.79) for lipegfilgrastim versus pegfilgrastim. The risk of severe neutropenia in cycles 2-4 was also significantly lower for lipegfilgrastim (risk ratio 0.45, 0.27, 0.75, respectively). No significant differences were found for febrile neutropenia and severe neutropenia in cycle 1. However, in cycles 2-4, lipegfilgrastim was associated with significant and clinically meaningful reductions in risk of severe neutropenia versus either pegfilgrastim or filgrastim. Conclusions Compared with pegfilgrastim or filgrastim, lipegfilgrastim has a statistically significantly lower absolute neutrophil count recovery time; however, differences in duration of severe neutropenia and bone pain were nonsignificant.
Keywords: Neutropenia; filgrastim; lipegfilgrastim; meta-analysis; pegfilgrastim.
Figures
References
-
- Crawford J, Dale DC, Lyman GH. Chemotherapy-induced neutropenia: risks, consequences, and new directions for its management. Cancer 2004; 100: 228–237. - PubMed
-
- Lyman GH, Dale DC, Crawford J. Incidence and predictors of low dose-intensity in adjuvant breast cancer chemotherapy: a nationwide study of community practices. J Clin Oncol 2003; 21: 4524–4531. - PubMed
-
- Lyman GH, Kuderer N, Greene J, et al. The economics of febrile neutropenia: implications for the use of colony-stimulating factors. Eur J Cancer 1998; 34: 1857–1864. - PubMed
-
- Webster J, Lyman GH. Use of G-CSF to sustain dose intensity in breast cancer patients receiving adjuvant chemotherapy: a pilot study. Cancer Control 1996; 3: 519–523. - PubMed
-
- Klastersky J, de Naurois J, Rolston K, et al. Management of febrile neutropaenia: ESMO Clinical Practice Guidelines. Ann Oncol 2016; 27: v111–v118. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources