Molecular mechanisms of signaling via the docosanoid neuroprotectin D1 for cellular homeostasis and neuroprotection
- PMID: 28615451
- PMCID: PMC5535015
- DOI: 10.1074/jbc.R117.783076
Molecular mechanisms of signaling via the docosanoid neuroprotectin D1 for cellular homeostasis and neuroprotection
Abstract
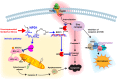
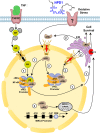
Docosahexaenoic acid, enriched in the brain and retina, generates docosanoids in response to disruptions of cellular homeostasis. Docosanoids include neuroprotectin D1 (NPD1), which is decreased in the CA1 hippocampal area of patients with early-stage Alzheimer's disease (AD). We summarize here how NPD1 elicits neuroprotection by up-regulating c-REL, a nuclear factor (NF)-κB subtype that, in turn, enhances expression of BIRC3 (baculoviral inhibitor of apoptosis repeat-containing protein 3) in the retina and in experimental stroke, leading to neuroprotection. Elucidating the mechanisms of action of docosanoids will contribute to managing diseases, including stroke, AD, age-related macular degeneration, traumatic brain injury, Parkinson's disease, and other neurodegenerations.
Keywords: NF-kB transcription factor; cell death; lipid signaling; neurodegenerative disease; retina.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures

References
-
- Selye H. (1998) A syndrome produced by diverse nocuous agents. 1936. J. Neuropsychiatry Clin. Neurosci. 10, 230–231 - PubMed
-
- Kolko M., Wang J., Zhan C., Poulsen K. A., Prause J. U., Nissen M. H., Heegaard S., and Bazan N. G. (2007) Identification of intracellular phospholipases A2 in the human eye: involvement in phagocytosis of photoreceptor outer segments. Invest. Ophthalmol. Vis. Sci. 48, 1401–1409 - PubMed
-
- LaVail M. M. (1980) Circadian nature of rod outer segment disc shedding in the rat. Invest. Ophthalmol. Vis. Sci. 19, 407–411 - PubMed
-
- Bok D. (1993) The retinal pigment epithelium: a versatile partner in vision. J. Cell Sci. Suppl. 17, 189–195 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

