Microbiota regulates visceral pain in the mouse
- PMID: 28629511
- PMCID: PMC5478269
- DOI: 10.7554/eLife.25887
Microbiota regulates visceral pain in the mouse
Abstract
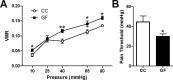
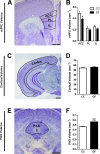
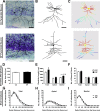
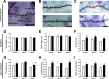
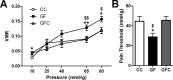
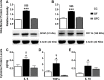
The perception of visceral pain is a complex process involving the spinal cord and higher order brain structures. Increasing evidence implicates the gut microbiota as a key regulator of brain and behavior, yet it remains to be determined if gut bacteria play a role in visceral sensitivity. We used germ-free mice (GF) to assess visceral sensitivity, spinal cord gene expression and pain-related brain structures. GF mice displayed visceral hypersensitivity accompanied by increases in Toll-like receptor and cytokine gene expression in the spinal cord, which were normalized by postnatal colonization with microbiota from conventionally colonized (CC). In GF mice, the volumes of the anterior cingulate cortex (ACC) and periaqueductal grey, areas involved in pain processing, were decreased and enlarged, respectively, and dendritic changes in the ACC were evident. These findings indicate that the gut microbiota is required for the normal visceral pain sensation.
Keywords: Irritable Bowel Syndrome; infectious disease; microbiology; microbiome; microbiota-gut-brain axis; mouse; neuroscience; periaqueductal gray; stress; toll-like receptor.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures



Similar articles
-
Disturbance of the gut microbiota in early-life selectively affects visceral pain in adulthood without impacting cognitive or anxiety-related behaviors in male rats.Neuroscience. 2014 Sep 26;277:885-901. doi: 10.1016/j.neuroscience.2014.07.054. Epub 2014 Aug 1. Neuroscience. 2014. PMID: 25088912
-
Berberine alleviates visceral hypersensitivity in rats by altering gut microbiome and suppressing spinal microglial activation.Acta Pharmacol Sin. 2021 Nov;42(11):1821-1833. doi: 10.1038/s41401-020-00601-4. Epub 2021 Feb 8. Acta Pharmacol Sin. 2021. PMID: 33558654 Free PMC article.
-
The Role of the Gastrointestinal Microbiota in Visceral Pain.Handb Exp Pharmacol. 2017;239:269-287. doi: 10.1007/164_2016_115. Handb Exp Pharmacol. 2017. PMID: 28035535 Review.
-
The gut microbiota as a key regulator of visceral pain.Pain. 2017 Apr;158 Suppl 1:S19-S28. doi: 10.1097/j.pain.0000000000000779. Pain. 2017. PMID: 27918315 Review. No abstract available.
-
Stress and the Microbiota-Gut-Brain Axis in Visceral Pain: Relevance to Irritable Bowel Syndrome.CNS Neurosci Ther. 2016 Feb;22(2):102-17. doi: 10.1111/cns.12490. Epub 2015 Dec 10. CNS Neurosci Ther. 2016. PMID: 26662472 Free PMC article. Review.
Cited by
-
Involvement of toll-like receptor 5 in mouse model of colonic hypersensitivity induced by neonatal maternal separation.World J Gastroenterol. 2022 Aug 7;28(29):3903-3916. doi: 10.3748/wjg.v28.i29.3903. World J Gastroenterol. 2022. PMID: 36157543 Free PMC article.
-
Gut-Brain Psychology: Rethinking Psychology From the Microbiota-Gut-Brain Axis.Front Integr Neurosci. 2018 Sep 11;12:33. doi: 10.3389/fnint.2018.00033. eCollection 2018. Front Integr Neurosci. 2018. PMID: 30271330 Free PMC article. Review.
-
Microbiome Influence in the Pathogenesis of Prion and Alzheimer's Diseases.Int J Mol Sci. 2019 Sep 23;20(19):4704. doi: 10.3390/ijms20194704. Int J Mol Sci. 2019. PMID: 31547531 Free PMC article. Review.
-
Resilience priming: Translational models for understanding resiliency and adaptation to early life adversity.Dev Psychobiol. 2019 Apr;61(3):350-375. doi: 10.1002/dev.21775. Epub 2018 Oct 11. Dev Psychobiol. 2019. PMID: 30311210 Free PMC article. Review.
-
Inflammasome Signaling Regulates the Microbial-Neuroimmune Axis and Visceral Pain in Mice.Int J Mol Sci. 2021 Aug 3;22(15):8336. doi: 10.3390/ijms22158336. Int J Mol Sci. 2021. PMID: 34361102 Free PMC article.
References
-
- Ait-Belgnaoui A, Han W, Lamine F, Eutamene H, Fioramonti J, Bueno L, Theodorou V. Lactobacillus farciminis treatment suppresses stress induced visceral hypersensitivity: a possible action through interaction with epithelial cell cytoskeleton contraction. Gut. 2006;55:1090–1094. doi: 10.1136/gut.2005.084194. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

