The Dual Activity Responsible for the Elongation and Branching of β-(1,3)-Glucan in the Fungal Cell Wall
- PMID: 28634239
- PMCID: PMC5478894
- DOI: 10.1128/mBio.00619-17
The Dual Activity Responsible for the Elongation and Branching of β-(1,3)-Glucan in the Fungal Cell Wall
Abstract
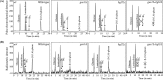
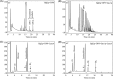
β-(1,3)-Glucan, the major fungal cell wall component, ramifies through β-(1,6)-glycosidic linkages, which facilitates its binding with other cell wall components contributing to proper cell wall assembly. Using Saccharomyces cerevisiae as a model, we developed a protocol to quantify β-(1,6)-branching on β-(1,3)-glucan. Permeabilized S. cerevisiae and radiolabeled substrate UDP-(14C)glucose allowed us to determine branching kinetics. A screening aimed at identifying deletion mutants with reduced branching among them revealed only two, the bgl2Δ and gas1Δ mutants, showing 15% and 70% reductions in the branching, respectively, compared to the wild-type strain. Interestingly, a recombinant Gas1p introduced β-(1,6)-branching on the β-(1,3)-oligomers following its β-(1,3)-elongase activity. Sequential elongation and branching activity of Gas1p occurred on linear β-(1,3)-oligomers as well as Bgl2p-catalyzed products [short β-(1,3)-oligomers linked by a linear β-(1,6)-linkage]. The double S. cerevisiae gas1Δ bgl2Δ mutant showed a drastically sick phenotype. An ScGas1p ortholog, Gel4p from Aspergillus fumigatus, also showed dual β-(1,3)-glucan elongating and branching activity. Both ScGas1p and A. fumigatus Gel4p sequences are endowed with a carbohydrate binding module (CBM), CBM43, which was required for the dual β-(1,3)-glucan elongating and branching activity. Our report unravels the β-(1,3)-glucan branching mechanism, a phenomenon occurring during construction of the cell wall which is essential for fungal life.IMPORTANCE The fungal cell wall is essential for growth, morphogenesis, protection, and survival. In spite of being essential, cell wall biogenesis, especially the core β-(1,3)-glucan ramification, is poorly understood; the ramified β-(1,3)-glucan interconnects other cell wall components. Once linear β-(1,3)-glucan is synthesized by plasma membrane-bound glucan synthase, the subsequent event is its branching event in the cell wall space. Using Saccharomyces cerevisiae as a model, we identified GH72 and GH17 family glycosyltransferases, Gas1p and Bgl2p, respectively, involved in the β-(1,3)-glucan branching. The sick phenotype of the double Scgas1Δ bgl2Δ mutant suggested that β-(1,3)-glucan branching is essential. In addition to ScGas1p, GH72 family ScGas2p and Aspergillus fumigatus Gel4p, having CBM43 in their sequences, showed dual β-(1,3)-glucan elongating and branching activity. Our report identifies the fungal cell wall β-(1,3)-glucan branching mechanism. The essentiality of β-(1,3)-glucan branching suggests that enzymes involved in the glucan branching could be exploited as antifungal targets.
Keywords: Aspergillus fumigatus; Saccharomyces cerevisiae; beta-glucan; cell wall; remodeling.
Copyright © 2017 Aimanianda et al.
Figures






References
-
- Mateos P, Domínguez A. 1991. Ultrastructure and cell wall composition in cell division cycle mutants of Schizosaccharomyces pombe deficient in septum formation. Antonie Van Leeuwenhoek 59:155–165. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

