The formation of lipid droplets favors intracellular Mycobacterium leprae survival in SW-10, non-myelinating Schwann cells
- PMID: 28636650
- PMCID: PMC5495515
- DOI: 10.1371/journal.pntd.0005687
The formation of lipid droplets favors intracellular Mycobacterium leprae survival in SW-10, non-myelinating Schwann cells
Abstract
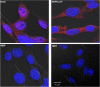
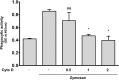
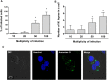
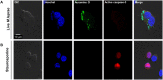
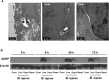
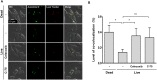
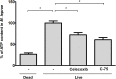
Leprosy is a chronic infectious disease that is caused by the obligate intracellular pathogen Mycobacterium leprae (M.leprae), which is the leading cause of all non-traumatic peripheral neuropathies worldwide. Although both myelinating and non-myelinating Schwann cells are infected by M.leprae in patients with lepromatous leprosy, M.leprae preferentially invades the non-myelinating Schwann cells. However, the effect of M.leprae infection on non-myelinating Schwann cells has not been elucidated. Lipid droplets (LDs) are found in M.leprae-infected Schwann cells in the nerve biopsies of lepromatous leprosy patients. M.leprae-induced LD formation favors intracellular M.leprae survival in primary Schwann cells and in a myelinating Schwann cell line referred to as ST88-14. In the current study, we initially characterized SW-10 cells and investigated the effects of LDs on M.leprae-infected SW-10 cells, which are non-myelinating Schwann cells. SW-10 cells express S100, a marker for cells from the neural crest, and NGFR p75, a marker for immature or non-myelinating Schwann cells. SW-10 cells, however, do not express myelin basic protein (MBP), a marker for myelinating Schwann cells, and myelin protein zero (MPZ), a marker for precursor, immature, or myelinating Schwann cells, all of which suggests that SW-10 cells are non-myelinating Schwann cells. In addition, SW-10 cells have phagocytic activity and can be infected with M. leprae. Infection with M. leprae induces the formation of LDs. Furthermore, inhibiting the formation of M. leprae-induced LD enhances the maturation of phagosomes containing live M.leprae and decreases the ATP content in the M. leprae found in SW-10 cells. These facts suggest that LD formation by M. leprae favors intracellular M. leprae survival in SW-10 cells, which leads to the logical conclusion that M.leprae-infected SW-10 cells can be a new model for investigating the interaction of M.leprae with non-myelinating Schwann cells.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Global leprosy update, 2015: time for action, accountability and inclusion. Wkly Epidemiol Rec. 2015. September 02;91(35):405–420. Epub 2016/09/07. . - PubMed
-
- Scollard DM. The biology of nerve injury in leprosy. Lepr Rev. 2008. September;79(3):242–253. Epub 2008/11/18. . - PubMed
-
- Rambukkana A, Zanazzi G, Tapinos N, Salzer JL. Contact-dependent demyelination by Mycobacterium leprae in the absence of immune cells. Science. 2002. May 03;296(5569):927–931. Epub 2002/05/04. doi: 10.1126/science.1067631 . - DOI - PubMed
-
- Rambukkana A. Mycobacterium leprae-induced demyelination: a model for early nerve degeneration. Curr Opin Immunol. 2004. August;16(4):511–518. Epub 2004/07/13. doi: 10.1016/j.coi.2004.05.021 . - DOI - PubMed
-
- Spierings E, De Boer T, Zulianello L, Ottenhoff TH. Novel mechanisms in the immunopathogenesis of leprosy nerve damage: the role of Schwann cells, T cells and Mycobacterium leprae. Immunol Cell Biol. 2000. August;78(4):349–355. Epub 2000/08/18. doi: 10.1046/j.1440-1711.2000.00939.x . - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

