Aberrant expression of kallikrein-related peptidase 7 is correlated with human melanoma aggressiveness by stimulating cell migration and invasion
- PMID: 28636767
- PMCID: PMC5623816
- DOI: 10.1002/1878-0261.12103
Aberrant expression of kallikrein-related peptidase 7 is correlated with human melanoma aggressiveness by stimulating cell migration and invasion
Abstract
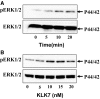
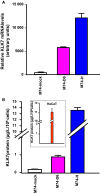
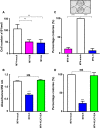
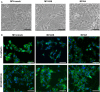
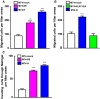
Members of the tissue kallikrein-related peptidase (KLK) family not only regulate several important physiological functions, but aberrant expression has also been associated with various malignancies. Clinically, KLKs have been suggested as promising biomarkers for diagnosis and prognosis in many types of cancer. As of yet, expression of KLKs and their role in skin cancers are, however, poorly addressed. Malignant melanoma is an aggressive disease associated with poor prognosis. Hence, diagnostic biomarkers to monitor melanoma progression are needed. Herein, we demonstrate that although mRNA of several KLKs are aberrantly expressed in melanoma cell lines, only the KLK7 protein is highly secreted in vitro. In line with these findings, ectopic expression of KLK7 in human melanomas and its absence in benign nevi were demonstrated by immunohistochemistry in vivo. Interestingly, overexpression of KLK7 induced a significant reduction in melanoma cell proliferation and colony formation. Moreover, KLK7 overexpression triggered an increase in cell motility and invasion associated with decreased expression of E-cadherin and an upregulation of MCAM/CD146. Our results demonstrate, for the first time, that aberrant KLK7 expression leads to a switch from proliferative to invasive phenotype, suggesting a potential role of KLK7 in melanoma progression. Thus, we hypothesize that KLK7 may represent a potential biomarker for melanoma progression.
Keywords: E-cadherin; invasion; kallikrein-related peptidase 7; melanoma; migration; proliferation.
© 2017 The Authors. Published by FEBS Press and John Wiley & Sons Ltd.
Figures


References
-
- French Public Health Code: Bioethical Law 2004‐800 of August 6, 2004 on bioethics. Journal official 182, Art. L. 1232‐1, Art. L. 1235‐2, Art. L. 1245‐2.
-
- Borgono CA and Diamandis EP (2004) The emerging roles of human tissue kallikreins in cancer. Nat Rev Cancer 4, 876–890. - PubMed
-
- Caubet C, Jonca N, Brattsand M, Guerrin M, Bernard D, Schmidt R, Egelrud T, Simon M, Serre G (2004) Degradation of corneodesmosome proteins by two serine proteases of the kallikrein family, SCTE/KLK5/hK5 and SCCE/KLK7/hK7. J Invest Dermatol. 122, 1235–1244. - PubMed
-
- Chung H, Hamza M, Oikonomopoulou K, Gratio V, Saifeddine M, Virca GD, Diamandis EP, Hollenberg MD, Darmoul D (2012) Kallikrein‐related peptidase signaling in colon carcinoma cells: targeting proteinase‐activated receptors. Biol Chem 393, 413–420. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

