Anti-vascular endothelial growth factor for diabetic macular oedema: a network meta-analysis
- PMID: 28639415
- PMCID: PMC6481463
- DOI: 10.1002/14651858.CD007419.pub5
Anti-vascular endothelial growth factor for diabetic macular oedema: a network meta-analysis
Update in
-
Anti-vascular endothelial growth factor for diabetic macular oedema: a network meta-analysis.Cochrane Database Syst Rev. 2018 Oct 16;10(10):CD007419. doi: 10.1002/14651858.CD007419.pub6. Cochrane Database Syst Rev. 2018. Update in: Cochrane Database Syst Rev. 2023 Jun 27;2023(6):CD007419. doi: 10.1002/14651858.CD007419.pub7. PMID: 30325017 Free PMC article. Updated.
Abstract
Background: Diabetic macular oedema (DMO) is a common complication of diabetic retinopathy. Antiangiogenic therapy with anti-vascular endothelial growth factor (anti-VEGF) modalities can reduce oedema and thereby improve vision and prevent further visual loss. These drugs have replaced laser photocoagulation as the standard of care for people with DMO.
Objectives: The 2014 update of this review found high-quality evidence of benefit with antiangiogenic therapy with anti-VEGF modalities, compared to laser photocoagulation, for the treatment of DMO.The objective of this updated review is to compare the effectiveness and safety of the different anti-VEGF drugs in preserving and improving vision and quality of life using network meta-analysis methods.
Search methods: We searched various electronic databases on 26 April 2017.
Selection criteria: We included randomised controlled trials (RCTs) that compared any anti-angiogenic drug with an anti-VEGF mechanism of action versus another anti-VEGF drug, another treatment, sham or no treatment in people with DMO.
Data collection and analysis: We used standard Cochrane methods for pair-wise meta-analysis and we augmented this evidence using network meta-analysis methods. We focused on the relative efficacy and safety of the three most commonly used drugs as interventions of direct interest for practice: aflibercept and ranibizumab, used on-label; and off-label bevacizumab.We collected data on three efficacy outcomes (gain of 15 or more Early Treatment Diabetic Retinopathy Study (ETDRS) letters; mean change in best-corrected visual acuity (BCVA); mean change in central retinal thickness (CRT)), three safety outcomes (all severe systemic adverse events (SSAEs); all-cause death; arterial thromboembolic events) and quality of life.We used Stata 'network' meta-analysis package for all analyses. We investigated the risk of bias of mixed comparisons based on the variance contribution of each study, having assigned an overall risk of bias to each study.
Main results: Twenty-four studies included 6007 participants with DMO and moderate vision loss, of which two studies randomised 265 eyes of 230 participants and one was a cross-over study on 56 participants (62 eyes) that was treated as a parallel-arm trial. Data were collected on drugs of direct interest from three studies on aflibercept (975 eyes), eight studies on bevacizumab (515 eyes), and 14 studies on ranibizumab (1518 eyes). As treatments of indirect interest or legacy treatment we included three studies on pegaptanib (541 eyes), five studies on ranibizumab plus prompt laser (557 eyes), one study on ranibizumab plus deferred laser (188 eyes), 13 studies on laser photocoagulation (936 eyes) and six studies on sham treatment (793 eyes).Aflibercept, bevacizumab and ranibizumab were all more effective than laser for improving vision by 3 or more lines after one year (high-certainty evidence). Approximately one in 10 people improve vision with laser, and about three in 10 people improve with anti-VEGF treatment: risk ratio (RR) versus laser 3.66 (95% confidence interval (CI) 2.79 to 4.79) for aflibercept; RR 2.47 (95% CI 1.81 to 3.37) for bevacizumab; RR 2.76 (95% CI 2.12 to 3.59) for ranibizumab. On average there was no change in visual acuity (VA) with laser after one year, compared with a gain of 1 or 2 lines with anti-VEGF treatment: laser versus aflibercept mean difference (MD) -0.20 (95% CI -0.22 to -0.17) logMAR; versus bevacizumab MD -0.12 (95% CI -0.15 to -0.09) logMAR; versus ranibizumab MD -0.12 (95% CI -0.14 to -0.10) logMAR. The certainty of the evidence was high for the comparison of aflibercept and ranibizumab with laser and moderate for bevacizumab comparison with laser due to inconsistency between the indirect and direct evidence.People receiving ranibizumab were less likely to gain 3 or more lines of VA at one year compared with aflibercept: RR 0.75 (95% CI 0.60 to 0.94), moderate-certainty evidence. For every 1000 people treated with aflibercept, 92 fewer would gain 3 or more lines of VA at one year if treated with ranibizumab (22 to 148 fewer). On average people receiving ranibizumab had worse VA at one year (MD 0.08 logMAR units, 95% CI 0.05 to 0.11), moderate-certainty evidence; and higher CRT (MD 39 µm, 95% CI 2 µm to 76 µm; low-certainty evidence). Ranibizumab and bevacizumab were comparable with respect to aflibercept and did not differ in terms of VA: RR of gain of 3 or more lines of VA at one year 1.11 (95% CI 0.87 to 1.43), moderate-certainty evidence, and difference in change in VA was 0.00 (95% CI -0.02 to 0.03) logMAR, moderate-certainty evidence. CRT reduction favoured ranibizumab by -29 µm (95% CI -58 µm to -1 µm, low-certainty evidence). There was no evidence of overall statistical inconsistency in our analyses.The previous version of this review found moderate-certainty evidence of good safety of antiangiogenic drugs versus control. This update used data at the longest available follow-up (one or two years) and found that aflibercept, ranibizumab and bevacizumab do not differ regarding systemic serious adverse events (SSAEs) (moderate- or high-certainty evidence). However, risk of bias was variable, loop inconsistency could be found and estimates were not precise enough on relative safety regarding less frequent events such as arterial thromboembolic events or death (low- or very low-certainty evidence).Two-year data were available and reported in only four RCTs in this review. Most industry-sponsored studies were open-label after one year. One large publicly-funded study compared the three drugs at two years and found no difference.
Authors' conclusions: Anti-VEGF drugs are effective at improving vision in people with DMO with three to four in every 10 people likely to experience an improvement of 3 or more lines VA at one year. There is moderate-certainty evidence that aflibercept confers some advantage over ranibizumab and bevacizumab in people with DMO at one year in visual and anatomic terms. Relative effects among anti-VEGF drugs at two years are less well known, since most studies were short term. Evidence from RCTs may not apply to real-world practice, where people in need of antiangiogenic treatment are often under-treated and under-monitored.We found no signals of differences in overall safety between the three antiangiogenic drugs that are currently available to treat DMO, but our estimates are imprecise for cardiovascular events and death.
Conflict of interest statement
Gianni Virgili: none known Mariacristina Parravano received payment for participating on the Advisory Board for Allergan, Bayer and Novartis. Jennifer Evans: none known Iris Gordon: none known Ersilia Lucenteforte: none known
Figures
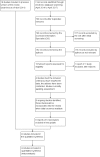









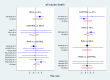


Update of
-
Anti-vascular endothelial growth factor for diabetic macular oedema.Cochrane Database Syst Rev. 2014 Oct 24;(10):CD007419. doi: 10.1002/14651858.CD007419.pub4. Cochrane Database Syst Rev. 2014. Update in: Cochrane Database Syst Rev. 2017 Jun 22;6:CD007419. doi: 10.1002/14651858.CD007419.pub5. PMID: 25342124 Updated.
References
References to studies included in this review
-
- Ahmadieh H, Ramezani A, Shoeibi N, Bijanzadeh B, Tabatabaei A, Azarmina M, et al. Intravitreal bevacizumab with or without triamcinolone for refractory diabetic macular edema; a placebo‐controlled, randomized clinical trial. Graefe's Archive for Clinical and Experimental Ophthalmology 2008;246(4):483‐9. - PubMed
-
- Michaelides M, Kaines A, Hamilton RD, Fraser‐Bell S, Rajendram R, Quhill F, et al. A prospective randomized trial of intravitreal bevacizumab or laser therapy in the management of diabetic macular edema (BOLT study) 12‐month data: report 2. Ophthalmology 2010;117(6):1078‐86. - PubMed
- Rajendram R, Fraser‐Bell S, Kaines A, Michaelides M, Hamilton RD, Esposti SD, et al. A 2‐year prospective randomized controlled trial of intravitreal bevacizumab or laser therapy (BOLT) in the management of diabetic macular edema: 24‐month data: report 3. Archives of Ophthalmology 2012;130(8):972‐9. - PubMed
- Sivaprasad S, Crosby‐Nwaobi R, Esposti S, Peto T, Rajendram R, Michaelides M, et al. Structural and functional measures of efficacy in response to bevacizumab monotherapy in diabetic macular oedema: exploratory analyses of the BOLT Study (Report 4). PloS ONE 2013;8(8):e72755. - PMC - PubMed
-
- Do DV, Nguyen QD, Boyer D, Schmidt‐Erfurth U, Brown DM, Vitti R, et al. One‐year outcomes of the DA VINCI Study of VEGF Trap‐Eye in eyes with diabetic macular edema. Ophthalmology 2012;119(8):1658‐65. - PubMed
- Do DV, Schmidt‐Erfurth U, Gonzalez VH, Gordon CM, Tolentino M, Berliner AJ, et al. The DAVINCI Study: phase 2 primary results of VEGF Trap‐Eye in patients with diabetic macular edema. Ophthalmology 2011;118(9):1819‐26. - PubMed
-
- Anonymous. Erratum: Patient‐reported visual function outcomes improve after ranibizumab treatment in patients with vision impairment due to diabetic macular edema: Randomized clinical trial (JAMA Ophthalmology (2013) 131:10 (1339‐1347) DOI: 10.1001/jamaophthalmol.2013.4592). JAMA Ophthalmology 2013;131(12):1652. - PubMed
- Bressler SB, Qin H, Beck RW, Chalam KV, Kim JE, Melia M, et al. Factors associated with changes in visual acuity and central subfield thickness at 1 year after treatment for diabetic macular edema with ranibizumab. Archives of Ophthalmology 2012;130(9):1153‐61. - PMC - PubMed
- Bressler SB, Qin H, Melia M, Bressler NM, Beck RW, Chan CK, et al. Exploratory analysis of the effect of intravitreal ranibizumab or triamcinolone on worsening of diabetic retinopathy in a randomized clinical trial. JAMA Ophthalmology 2013;131(8):1033‐40. - PMC - PubMed
- Dewan V, Lambert D, Edler J, Kymes S, Apte RS. Cost‐effectiveness analysis of ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology 2012;119(8):1679‐84. - PMC - PubMed
- Diabetic Retinopathy Clinical Research Network, Elman MJ, Aiello LP, Beck RW, Bressler NM, Bressler SB, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology 2010;117(6):1064–77. - PMC - PubMed
- Elman MJ, Bressler NM, Qin H, Beck RW, Ferris FL 3rd, Friedman SM, et al. Expanded 2‐year follow‐up of ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology 2011;118(4):609‐14. - PMC - PubMed
- Elman MJ, Qin H, Aiello LP, Beck RW, Bressler NM, Ferris FL, et al. Intravitreal ranibizumab for diabetic macular edema with prompt versus deferred laser treatment: three‐year randomized trial results. Ophthalmology 2012;119(11):2312‐8. - PMC - PubMed
References to studies excluded from this review
-
- Ahmadieh H, Nourinia R, Hafezi‐Moghadam A. Intravitreal fasudil combined with bevacizumab for persistent diabetic macular edema. JAMA Ophthalmology 2013;131(7):923‐4. - PubMed
-
- CRFB002DFR08. Open‐label, multicenter, study of the efficacy and safety of Lucentis® (ranibizumab 0.5 mg) in diabetic patients presenting a visual impairment due to diabetic macular edema in current medical practice (LUDIC). Novartis clinical trial results database. www.novctrd.com/CtrdWeb/displaypdf.nov?trialresultid=9803 (accessed 28 May 2014).
-
- CRFB002DGB14. RELIGHT ‐ Ranibizumab treatment of diabetic macular oEdema with bimonthLy monItorinG after a pHase of initial Treatment. A UK, 18‐month, prospective, open‐label, multicentre, single‐arm Phase IIIb study, with 12‐month primary endpoint, assessing the efficacy and safety of Ranibizumab in patients with visual impairment. Novartis clinical trial results database www.novctrd.com/ctrdWebApp/clinicaltrialrepository/public/products.jsp?d... (accessed 28 May 2014).
-
- CRFB002DNO02. An open‐label, prospective, multicenter, uncontrolled, Proof of concept study assessing the efficacy of Lucentis (ranibizumab) administered by an individualized “treat and extend” dosing regimen in patients with visual impairment due to dIabetic macular edema. Novartis clinical trial results database. www.novctrd.com/CtrdWeb/displaypdf.nov?trialresultid=10423 (accessed 28 May 2014).
References to studies awaiting assessment
-
- Chen ZX, Fu JS, Song W, Wang CX, Zhang YL. Effect analysis of ranibizumab with laser photocoagulation therapy for diabetic macular edema. International Eye Science 2016;16(4):706‐8.
-
- Huang JD, Song ZY. Clinical study of grid pattern laser photocoagulation with ranibizumab for diabetic macular edema. International Eye Science 2016;16(3):493‐5.
-
- Jovanović S, Čanadanović V, Sabo A, Grgić Z, Mitrović M, Rakić D. Intravitreal bevacizumab injection alone or combined with macular photocoagulation compared to macular photocoagulation as primary treatment of diabetic macular edema. Vojnosanitetski Pregled 2015;72(10):876‐82. - PubMed
-
- NCT00387582. Efficacy study of lucentis in the treatment of diabetic macular edema ‐ a phase II, single center, randomized study to evaluate the efficacy of ranibizumab versus focal laser treatment in subjects with diabetic macular edema [Lucentis in the treatment of macular edema ‐ a phase II, single center, randomized study to evaluate the efficacy of ranibizumab versus focal laser treatment in subjects with diabetic macular edema]. clinicaltrials.gov/show/NCT00387582 (first received 11 October 2006).
References to ongoing studies
-
- ChiCTR‐TRC‐12002417. A randomized controlled trial to compare the efficacy and safety of 1) macular laser vs. 2) repeated intravitreal bevacizumab vs. 3) combined repeated intravitreal bevacizumab with macular laser for diabetic macular edema. www.chictr.org.cn/hvshowproject.aspx?id=3319 (accessed 17 September 2014).
-
- NCT01635790. Comparing the effectiveness and costs of bevacizumab to ranibizumab in patients with diabetic macular edema (BRDME) (BRDME). clinicaltrials.gov/ct2/show/NCT01635790 (first received 28 June 2012). - PMC - PubMed
-
- NCT02194634. Safety and efficacy study of conbercept in diabetic macular edema (DME) (Sailing). clinicaltrials.gov/ct2/show/NCT02194634 (first received 14 July 2014).
-
- NCT02259088. A 12‐month, randomized, efficacy and safety study of 0.5 mg ranibizumab vs laser in Chinese DME patients [A 12‐month, randomized, double‐masked, multicenter, laser‐controlled phase III study assessing the efficacy and safety of 0.5 mg ranibizumab dosed PRN in subjects with visual impairment due to diabetic macular edema in Chinese patients]. clinicaltrials.gov/ct2/show/NCT02259088 (first received 3 October 2014).
-
- NCT02348918. A phase 2 randomized, controlled, double‐masked, multicenter clinical trial designed to evaluate the safety and exploratory efficacy of Luminate® (ALG‐1001) as compared to Avastin® and focal laser photocoagulation in the treatment of diabetic macular edema. clinicaltrials.gov/ct2/show/NCT02348918 (first received 12 January 2015).
Additional references
-
- Aiello LP. Angiogenic pathways in diabetic retinopathy. New England Journal of Medicine 2005;353(8):839‐41. - PubMed
-
- Antcliff RJ, Marshall J. The pathogenesis of edema in diabetic maculopathy. Seminars in Ophthalmology 1999;14(4):223‐32. - PubMed
-
- Arevalo JF, Lasave AF, Wu L, Diaz‐Llopis M, Gallego‐Pinazo R, Alezzandrini AA, et al. Intravitreal bevacizumab plus grid laser photocoagulation or intravitreal bevacizumab or grid laser photocoagulation for diffuse diabetic macular edema: results of the Pan‐american Collaborative Retina Study Group at 24 months. Retina 2013;33(2):403‐13. - PubMed
-
- Banfi R, Attanasio F, Palazzi N, Colombini S, Falai T, Cecchi M, et al. Bevacizumab versus ranibizumab: why are we not playing the joker?. International Journal of Clinical Pharmacy 2013;35(4):507‐9. - PubMed
References to other published versions of this review
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

